Aspectrum of cutaneous reactions has been reported worldwide by recipients of COVID-19 vaccines. Regarding reactions to mRNA COVID-19 vaccines, a registry-based study of 414 cases with cutaneous reactions to vaccines showed that delayed large local reactions were most common, followed by local injection site reactions, urticarial eruptions, and morbilliform eruptions.1 Less commonly reported were pernio/chilblains, cosmetic filler reactions, zoster, herpes simplex flares, and pityriasis rosea (PR)-like reactions.1
Rare cutaneous reactions to viral vector COVID-19 vaccines have also been reported.2,3 In Brazil, where the viral vector-based Oxford-AstraZeneca vaccine was the most widely used vaccine, the most common cutaneous reactions were injection site reactions, acute urticaria, and morbilliform rash. More rarely reported were lichen planus, purpura/vasculitis, erythroderma, and fixed drug eruption.2
We describe three cases of PR and PR-like eruption (PR-LE) that emerged after COVID-19 vaccinations. Two of the patients received Pfizer-BioNTech, an mRNA vaccine, while the third was administered Oxford-AstraZeneca, a viral vector vaccine.
Case Reports
Case One
A 19-year-old young man with no previously known health issues presented with a two-month history of pruritic, painless erythematous skin eruption over the trunk and the proximal extremities. The eruption was progressive and persistent. There was no associated systemic symptom or involvement of the scalp, nails, or mucus membranes. The patient also denied a history of recent dietary changes or new medication intake, but said that the symptoms began one week after receiving the first dose of Pfizer-BioNTech, an mRNA COVID-19 vaccine. Prior to the current presentation, he was treated with oral antihistamine and topical steroids, with no significant benefit.
Skin examination [Figure 1] showed erythematous, non-tender scaly papules and plaques that followed the skin tension lines over the lateral sides of the chest and coalesced in a reticular pattern over the lower back and abdomen. There was no petechial rash or wheals, and clinical examination revealed no evidence of a herald patch.
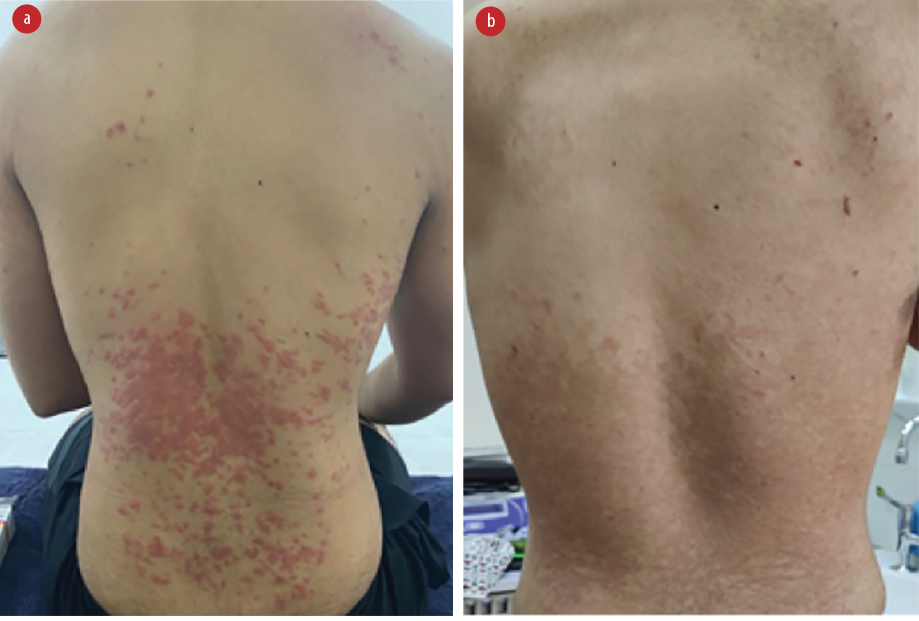 Figure 1: (a) On presentation, erythematous papules and plaques were seen coalescing over the trunk. (b) Two weeks into the treatment, the lesions are clearing, albeit with persistent reticular post-inflammatory hyperpigmentation.
Figure 1: (a) On presentation, erythematous papules and plaques were seen coalescing over the trunk. (b) Two weeks into the treatment, the lesions are clearing, albeit with persistent reticular post-inflammatory hyperpigmentation.
Skin biopsy microscopy [Figure 2] revealed spongiotic dermatitis with mild parakeratosis. There were few collections of inflammatory cells in the epidermis. The dermis showed perivascular and periadnexal inflammation, mostly lymphocytes. The features were suggestive of PR. Immunofluorescence was negative for the immunoglobulins IgA, IgM, IgG, and complement component C3. Serology for human herpesviruses 6 and 7 (HHV6/7) was not performed.
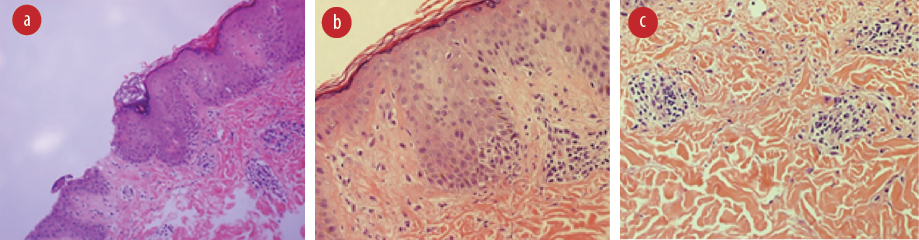 Figure 2: Hematoxylin and eosin stained skin biopsy showing (a) spongiosis and parakeratosis, magnification = 44 ×; (b) minimal inflammation in the epidermis, magnification = 10 ×; and
Figure 2: Hematoxylin and eosin stained skin biopsy showing (a) spongiosis and parakeratosis, magnification = 44 ×; (b) minimal inflammation in the epidermis, magnification = 10 ×; and
(c) periadnexal and perivascular lymphocytic infiltrate, magnification = 10 ×.
A course of oral azithromycin 500 mg once daily for three days and topical corticosteroids was prescribed. Progressive clearance of the eruption was noted after two weeks [Figure 1].
Case Two
A woman in her thirties with no previously known health issues presented with one week history of an erythematous pruritic skin eruption on the trunk, arms, and axillae. There were no associated constitutional symptoms. There was no mucosal or palmoplantar involvement. The patient denied any new medication intake. The eruptions had begun two weeks after receiving the first dose of Pfizer-BioNTech mRNA COVID-19 vaccine.
Skin examination showed erythematous confluent targetoid lesions with collarette scales on both forearms, arms, and trunk. There was a large annular erythematous plaque with central clearing and collarette scaling located on the left axilla, consistent with the herald patch. No petechial rash or wheal was noted [Figure 3].
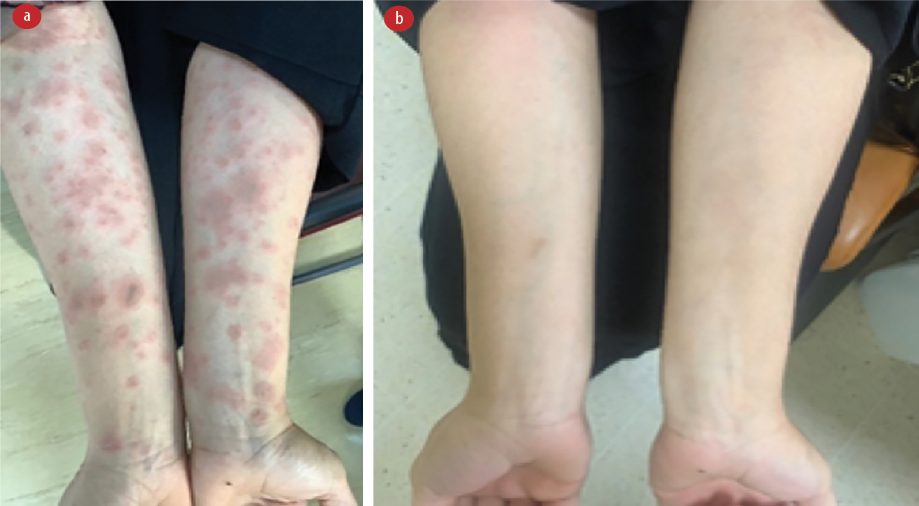 Figure 3: (a) On presentation, erythematous confluent targetoid lesions with collarette scales are seen consistent with herald patch formation. (b) After three weeks of treatment with azithromycin and corticosteroid, the rashes have cleared.
Figure 3: (a) On presentation, erythematous confluent targetoid lesions with collarette scales are seen consistent with herald patch formation. (b) After three weeks of treatment with azithromycin and corticosteroid, the rashes have cleared.
A course of oral azithromycin 500 mg for three days and topical corticosteroids were prescribed, and the eruption cleared in three weeks [Figure 4]. The patient subsequently took the second dose of the same vaccine, and a milder eruption occurred, which cleared spontaneously.
 Figure 4: Hematoxylin and eosin stained skin biopsy showing spongiosis (red arrows) with perivascular lymphocytic infiltrate (blue arrows), magnification = 4 ×.
Figure 4: Hematoxylin and eosin stained skin biopsy showing spongiosis (red arrows) with perivascular lymphocytic infiltrate (blue arrows), magnification = 4 ×.
Case Three
A man in his seventies with a history of hand dermatitis (though well-controlled by topical therapy) presented with two months history of new-onset pruritic eruption. It started on the trunk and then spread to the extremities. There were no preceding viral illness symptoms or recent exposure to new medications. Three weeks before the eruption, he had received his second dose of Oxford-AstraZeneca COVID-19 vaccine (a viral vector vaccine that encodes SARS-CoV-2 spike protein). The patient reported that the first dose had been uneventful.
Skin examination revealed eczematous oval-shaped plaques following cleavage lines, especially on the back, forming a Christmas tree pattern [Figure 5]. Some of the plaques showed collarette scales. There were multiple annular plaques with a clearing center on the left shoulder, similar to a herald patch. Other eczematous plaques were scattered over the forehead, neck, and abdomen. No mucosal or palmoplantar involvement was noted.
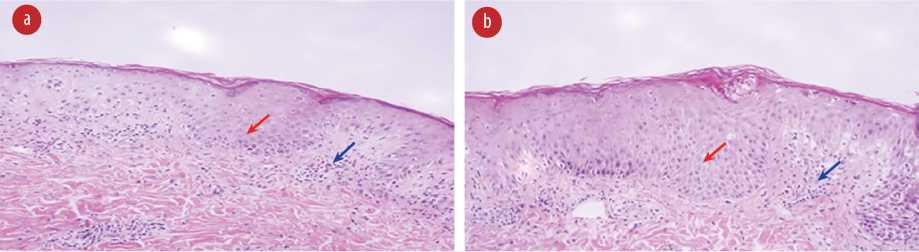 Figure 5: (a) On presentation, erythematous oval plaques are visible on the trunk along the cleavage lines. (b) After two weeks of treatment with azithromycin and antihistamine, improvement is noticeable.
Figure 5: (a) On presentation, erythematous oval plaques are visible on the trunk along the cleavage lines. (b) After two weeks of treatment with azithromycin and antihistamine, improvement is noticeable.
Skin biopsy showed focal parakeratosis and spongiosis with perivascular lymphocytic infiltrate with occasional neutrophils and red blood cell extravasation in the dermis [Figure 4].
Complete blood count showed no evidence of eosinophilia. HHV6/7 testing was not performed. The patient was treated with azithromycin and antihistamine, and a good response was noted in the two-week follow-up visit [Figure 5].
Discussion
PR is a papulosquamous disease, typically preceded by a primary solitary herald patch followed within days to weeks by the emergence of smaller scaly lesions along the skin tension lines.4 Possible triggers are bacterial or viral infections, medications, and vaccination. PR is mainly associated with endogenous systemic reactivation of
HHV6/7 infections.5,6
A wide spectrum of cutaneous reactions was observed after COVID-19 vaccinations worldwide. The reactions included hypersensitivity reactions of type I and type IV, functional angiopathies, autoimmune-mediated skin conditions, PR, and PR-LE. 7
In a registry-based study conducted in the USA between December 2020 and February 2021, 414 skin reactions to two mRNA-based COVID-19 vaccines—Moderna (83%) and Pfizer (17%)—were identified. Delayed local reactions were the most common, followed by urticarial and morbilliform eruptions. Among the patients who had reactions to the first dose, 43% experienced recurrence after the second dose, as did our patient in Case Two, who had also received an mRNA vaccine. Less commonly reported reactions include manifestations of herpes zoster, herpes simplex, and PR-LE. The occurrence of PR-LE was mentioned in one case after the first dose of Moderna, two cases after the first dose of Pfizer, and one case after the second dose of Pfizer.1 A middle-aged woman was reported to have developed PR 15 days after the second dose of the Oxford-AstraZeneca vaccine (viral vector-based), similar to our Case Three.8
A four-month-long nationwide study in Spain identified 405 instances of reaction after various types of COVID-19 vaccinations with the following break up: BNT162b2 (Pfizer-BioNTech; mRNA): 40.2%; mRNA-1273 (Moderna; mRNA): 36.3%, and AZD1222 (AstraZeneca; viral vector): 23.5%. PR-LE constituted 4.9% of all cutaneous reactions.9
A systematic review of 31 studies by Khan et al,3 included 111 patients (55.4% female) who developed PR or PR-LE after COVID-19 vaccination. Around 62% developed symptoms after the first dose and 38% after the second dose. Most had received the mRNA vaccines, Pfizer (35%) and Moderna (25%).3 The reported mean time of symptoms onset was 8.6 days, and recovery was 6.4 weeks.
Rare cases of PR and PR-LE have been reported after vaccinations against smallpox, tuberculosis, influenza, papillomaviruses, poliomyelitis, tetanus, diphtheria, pneumococcus, diphtheria-pertussis-tetanus, hepatitis B, and yellow fever. In such cases, the average time lapse between vaccination and eruption onset was 5–17 days, and the exanthema lasted 2–6 weeks. Differentiating between PR and PR-LE is difficult and virological investigations for HHV-6/7 reactivation have been performed only in a minority of cases.10–12 Cutaneous reactions post-COVID-19 vaccinations have been observed more in mRNA vaccines than in non-mRNA vaccines.13,14
Based on the criteria proposed by Drago et al,15,16 PR-LE is better viewed as a drug/vaccine hypersensitivity reaction that morphologically resembles PR, than as classic PR. PR-LE usually presents with severe itching and atypical morphology of the skin lesions, such as papules, vesicles, urticated plaques, purpura, and target lesions (erythema multiform-like). They present as large-sized lesions or as confluent plaques. Unusual distribution patterns of the skin lesions are more common in PR-LE, like an inverse pattern, prominent involvement of the skin folds, face, or greater involvement of limbs than the trunk. In addition, mucosal involvement is more prominent with PR-LE than with classic PR. The lack of herald patch is more prevalent in PR-LE. Regarding histopathology, PR-LE presents with a picture of interface dermatitis with eosinophils, while PR usually presents with parakeratosis, spongiosis, and lymphocytic infiltrate. PR-LE usually resolves faster if the culprit drug is stopped compared to PR.16
The pathogenetic mechanisms of post-vaccination PR and PR-LE are still unknown. One hypothesis is that the occurrence of high cytokine response to the vaccine leading to immune dysregulation and reactivation of latent viral infections such as HHV-6/7.10 PR-LE post-vaccination can be considered more of a delayed hypersensitivity reaction that might be explained by molecular mimicry mechanism between the host proteins and viral vector or a vaccine ingredient, resulting in skin eruption mediated by T cells.5,7
Case One in this series resembled PR but had atypical morphology and prominent itch. Hence, we diagnosed it as PR-LE. Case Two was more representative of classic PR, given its typical morphology with collarette scales and obvious herald patch, although the involvement of proximal upper limbs was not usual. Case Three had an atypical morphology of PR with typical pattern, possible herald patch and supporting histopathological features so we diagnosed it as PR. All three cases responded well to azithromycin, which was administered for its anti-inflammatory effect.
As per Drago et al,15 recurrences with the booster dose might not occur with PR. However, there was a recurrence in Case Two, which might point to more of a causal relationship. Our assumption is supported by a case from Turkey where a 45-year-old woman developed PR after receiving Sinovac-CoronaVac, a non-mRNA vaccine based on inactivated SARS-CoV-2. Her rash developed four days after receiving the first dose of the vaccine and was treated symptomatically with topical steroids and an oral antihistamine, and she had a recurrence after the second dose.17 In addition, although unusual, multiple recurrences of PR have been documented in the literature.3,18,19
Large-scale epidemiological studies are called for to further elucidate whether there is a relationship between vaccination regimens and the reactivation of latent viruses, and to assess whether such reactivation could be a coincidence or a consequence of the SARS-CoV-2 vaccines.
Conclusion
The three cases of PR and PR-LE reported here were possible cutaneous reactions to COVID-19 vaccination. Different types of COVID-19 vaccines—both mRNA and traditional vaccines—can produce such reactions. These reactions are rare, self-limiting, and only occasionally require treatment. We emphasize the necessity of vaccinating against COVID-19 while keeping close observation of any skin eruptions.
Disclosure
The authors declared no conflicts of interest. Written consent was obtained from all three patients.
references
- 1. McMahon DE, Amerson E, Rosenbach M, Lipoff JB, Moustafa D, Tyagi A, et al. Cutaneous reactions reported after Moderna and Pfizer COVID-19 vaccination: a registry-based study of 414 cases. J Am Acad Dermatol 2021 Jul;85(1):46-55.
- 2. Seque CA, Enokihara MMSS, Nascimento MM, Porro AM, Tomimori J. Rare cutaneous reactions after ChAdOx1 (Oxford-AstraZeneca) vaccine: 12 case series from Brazil. J Eur Acad Dermatol Venereol 2022 Aug;36(8):e601-e603.
- 3. Khan I, Elsanousi AA, Shareef AM, Tebha SS, Arif A, Gul S. Manifestation of pityriasis rosea and pityriasis rosea-like eruptions after covid-19 vaccine: a systematic review. Immun Inflamm Dis 2023 Apr;11(4):e804.
- 4. Chuh A, Lee A, Zawar V, Sciallis G, Kempf W. Pityriasis rosea–an update. Indian J Dermatol Venereol Leprol 2005 Sep-Oct;71(5):311-315.
- 5. Drago F, Broccolo F, Rebora A. Pityriasis rosea: an update with a critical appraisal of its possible herpesviral etiology. J Am Acad Dermatol 2009 Aug;61(2):303-318.
- 6. Watanabe T, Kawamura T, Jacob SE, Aquilino EA, Orenstein JM, Black JB, et al. Pityriasis rosea is associated with systemic active infection with both human herpesvirus-7 and human herpesvirus-6. J Invest Dermatol 2002 Oct;119(4):793-797.
- 7. Gambichler T, Boms S, Susok L, Dickel H, Finis C, Abu Rached N, et al. Cutaneous findings following COVID-19 vaccination: review of world literature and own experience. J Eur Acad Dermatol Venereol 2022 Feb;36(2):172-180.
- 8. Pedrazini MC, da Silva MH. Pityriasis rosea-like cutaneous eruption as a possible dermatological manifestation after Oxford-AstraZeneca vaccine: case report and brief literature review. Dermatol Ther 2021 Nov;34(6):e15129.
- 9. Català A, Muñoz-Santos C, Galván-Casas C, Roncero Riesco M, Revilla Nebreda D, Solá-Truyols A, et al. Cutaneous reactions after SARS-CoV-2 vaccination: a cross-sectional Spanish nationwide study of 405 cases. Br J Dermatol 2022 Jan;186(1):142-152.
- 10. Drago F, Ciccarese G, Javor S, Parodi A. Vaccine-induced pityriasis rosea and pityriasis rosea-like eruptions: a review of the literature. J Eur Acad Dermatol Venereol 2016 Mar;30(3):544-545.
- 11. Drago F, Ciccarese G, Rebora A, Parodi A. Pityriasis rosea following human papillomavirus vaccination. Braz J Infect Dis 2015 Mar-Apr;19(2):224-225.
- 12. Björnberg A, Hellgren L. Pityriasis rosea. A statistical, clinical, and laboratory investigation of 826 patients and matched healthy controls. Acta Derm Venereol Suppl (Stockh) 1962;42(Suppl 50):1-68.
- 13. Qaderi K, Golezar MH, Mardani A, Mallah MA, Moradi B, Kavoussi H, et al. Cutaneous adverse reactions of COVID-19 vaccines: a systematic review. Dermatol Ther 2022 May;35(5):e15391.
- 14. Sun Q, Fathy R, McMahon DE, Freeman EE. Covid-19 vaccines and the skin. Dermatol Clin 2021 Oct;39(4):653-673.
- 15. Drago F, Broccolo F, Ciccarese G. Pityriasis rosea, pityriasis rosea–like eruptions, and herpes zoster in the setting of COVID-19 and COVID-19 vaccination. Clin Dermatol 2022;40(5):586-590.
- 16. Drago F, Ciccarese G, Parodi A. Pityriasis rosea and pityriasis rosea-like eruptions: how to distinguish them? JAAD Case Rep 2018 Sep;4(8):800-801.
- 17. Akdaş E, İlter N, Öğüt B, Erdem Ö. Pityriasis rosea following coronavac COVID-19 vaccination: a case report. J Eur Acad Dermatol Venereol 2021 Aug;35(8):e491-e493.
- 18. Drago F, Ciccarese G, Rebora A, Parodi A. Relapsing pityriasis rosea. Dermatology 2014;229(4):316-318.
- 19. Sankararaman S, Velayuthan S. Multiple recurrences in pityriasis rosea - a case report with review of the literature. Indian J Dermatol 2014 May;59(3):316.