Median arcuate ligament syndrome (MALS) is a rare entity. First described by Harjola in 1963,1 MALS is characterized by the clinical triad of postprandial abdominal pain, weight loss, and epigastric bruit.1 The symptoms are due to the combined effects of celiac artery compression and irritation of the celiac plexus/ganglion by the MAL. Though celiac artery compression had been described as early as 1917, it took decades to understand its clinical implications.2 Being a diagnosis of exclusion, the possibility of MALS is considered after more common conditions have been excluded. Multiple imaging modalities can serve as diagnostic tools, including computed tomography angiography (CTA), magnetic resonance angiogram (MRA), Doppler ultrasonography, and selective angiography. Definitive management includes surgery involving either open or laparoscopic division of MAL and excision of celiac plexus.
Case Report
A 28-year-old man was referred to the surgical department from the gastroenterology clinic with a suspected diagnosis of MALS. He had been suffering from postprandial abdominal pain and bloating for the past two years. He also had nausea, vomiting, anorexia, and weight loss. Blood investigations and ultrasound scan of the abdomen yielded normal results. Upper gastrointestinal (GI) endoscopy showed mild gastritis and duodenitis. Computed tomographic enterography results were normal. Color Doppler ultrasound of the abdomen showed narrowing of the celiac trunk at its origin with post-stenotic dilatation. Finally, CTA was carried out which confirmed focal narrowing in the celiac ostium with dilatation of the celiac trunk distal to this stenotic segment, suggestive of MALS [Figures 1 and 2]. Upon confirmation of MALS, the patient was planned for laparoscopic division.
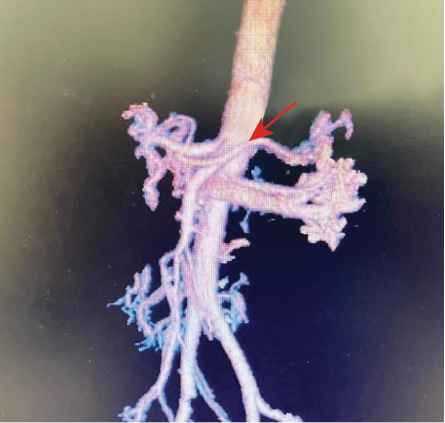 Figure 1: CT angiogram showing focal narrowing in the celiac ostium with post-stenotic dilatation.
Figure 1: CT angiogram showing focal narrowing in the celiac ostium with post-stenotic dilatation.
 Figure 2: CT angiogram: side view showing narrowing of the celiac ostium with post-
Figure 2: CT angiogram: side view showing narrowing of the celiac ostium with post-
stenotic dilatation.
The patient was placed in reverse Trendelenburg position with legs apart and the surgeon stood between his legs. Pneumoperitoneum was created by Veress needle and carbon dioxide was insufflated to a pressure of 13 mm Hg. We used ‘5-port technique’ which includes one camera port, two working ports, and two assistant ports. A 12 mm visiport was inserted just above the umbilicus to enter into the abdominal cavity (20 cm caudal from the xiphisternum allowed better visualization and accessibility to the aorta and the MAL). A 5 mm epigastric port inserted through which a snake liver retractor was passed to lift the left lobe of liver. Two working ports 5 mm and 10 mm were inserted in the right and left midclavicular lines in the upper abdomen. Another 5 mm assistant port was inserted in the left anterior axillary line for retraction.
The surgery began with right crural dissection. The gastrohepatic ligament was opened starting at pars flaccida [Figure 3]. The right phrenoesophageal membrane and the underneath fibers of the right crus of the diaphragm were dissected and mobilized from the esophagus.
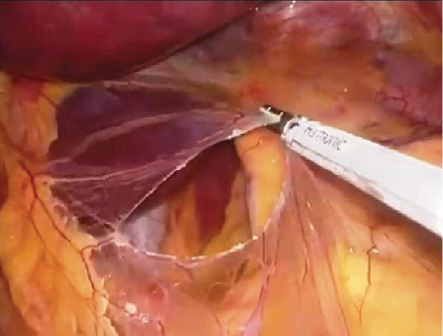 Figure 3: Intraoperative image of pars
Figure 3: Intraoperative image of pars
flaccida dissection.
The left gastric artery was isolated and hooked with an infant feeding tube and tractioned downwards [Figure 4a]. The artery was traced till its origin from the celiac trunk. The celiac trifurcation was identified and the dissection carried out along the origin of the left gastric artery, splenic artery and common hepatic artery, [Figure 4b]. The surrounding 5 cm of fibrofatty tissue was cleared, which helped identify the origin of the celiac trunk. The celiac trunk was followed till its origin from the aorta. The celiac plexus of nerves surrounding the celiac trunk were dissected and excised.
 Figure 4: (a) Hooking of the left gastric artery, (b) celiac trifurcation after fatty dissection, (c) and lift test of median arcuate ligament (MAL) confirming complete encasement of aorta. (d) division of MAL and the aorta partly cleared of encasement.
Figure 4: (a) Hooking of the left gastric artery, (b) celiac trifurcation after fatty dissection, (c) and lift test of median arcuate ligament (MAL) confirming complete encasement of aorta. (d) division of MAL and the aorta partly cleared of encasement.
The MAL was identified. Lift test confirmed complete encasement of aorta and compression of celiac trunk [Figure 4c]. The ligament was divided with the help of ultracision forceps and about 5 cm of adjacent aorta was cleared of the ligament [Figure 4d]. After the release of MAL, celiac trunk was traced up to the aorta which showed a uniform course without stenosis or kinking. The operative time was 90 minutes with minimal blood loss.
Postoperatively, the patient recovered well. On the same day, he was mobilized and fed orally. He was discharged satisfactorily on day two. On follow-up after four weeks, he was symptom-free and had gained 2 kg weight. He is on a regular follow-up.
Discussion
MALS is a rare entity. The normal anatomy of MAL and compression of the celiac trunk is shown in Figure 5. The aorta traverses through the chest over the vertebral bodies to enter the abdomen via the diaphragmatic opening. The MAL is a fibrous structure formed by right and left crura of the diaphragm that wraps around the aorta at the diaphragmatic opening.3 The celiac artery is the first large branch of the aorta that enters the abdomen. The MALS can occur either due to the celiac artery originating higher or to abnormal location of MAL.4
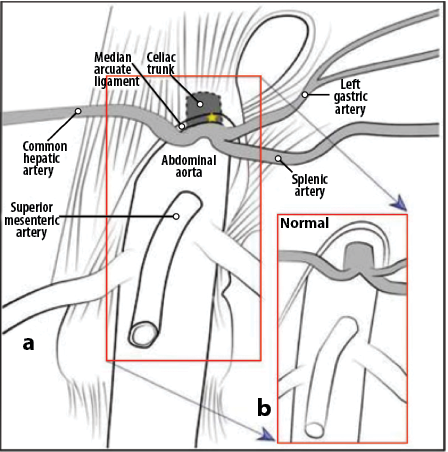 Figure 5: (a) Normal median arcuate ligament anatomy and (b) celiac trunk compression.
Figure 5: (a) Normal median arcuate ligament anatomy and (b) celiac trunk compression.
Being a diagnosis of exclusion, MALS should be considered after more common conditions are excluded.5 Epigastric bruit is seen in 85% of MALS cases, though our patient did not present with it. Interestingly, 30% of healthy adults also present with epigastric bruit.6,7
Women aged 20–60 years with thin body habitus are more vulnerable to MALS. The female to male ratio is reported to be 3:1.5 The reason for female predominance is unclear but may partly be due to the greater prevalence of cephalic origin of the celiac artery in females.6
The presenting symptoms of MALS are attributed to the combined effects of celiac artery compression and irritation of celiac plexus/ganglion, along with splanchnic vasoconstriction.5,7–10 Downward displacement with post-stenotic dilatation of the celiac artery during expiration is indicative of MALS.11
The definitive management is surgical (either open or laparoscopic) division of MAL with excision of the celiac plexus. Other options include endovascular stenting, bypass grafting, and percutaneous transluminal angioplasty. Laparoscopic division of MAL with excision of coeliac plexus typically yields the best results. Revascularization procedures have the drawbacks of lifelong blood-thinner and long-term follow-ups to assess the stent patency, of which the patients should be made aware.7–10
Conclusion
Laparoscopic release of the MAL with excision of the celiac plexus provides sustainable symptom relief in patients with MALS. It is an optimal, safe, and early-recovery procedure for this uncommon condition.
Disclosure
The authors declared no conflicts of interest. The patient was not reachable hence the clearance was taken from the ethical committee chairman.
references
- 1. Harjola PT. A rare obstruction of the coeliac artery. Report of a case. Ann Chir Gynaecol Fenn 1963;52:547-550.
- 2. Lipshutz B. A composite study of the coeliac axis artery. Ann Surg 1917 Feb;65(2):159-169.
- 3. Bayat I, Wang J, Ho P, Bird D. Intravascular ultrasound-guided laparoscopic division of the median arcuate ligament. Journal of Vascular Surgery Cases, Innovations and Techniques 2020 Mar 1;6(1):147-151.
- 4. Williams S, Gillespie P, Little JM. Celiac axis compression syndrome: factors predicting a favorable outcome. Surgery 1985 Nov;98(5):879-887.
- 5. Santos GM, Viarengo LM, Oliveira MD. Celiac artery compression: Dunbar syndrome. J Vasc Bras 2019 May;18:e20180094.
- 6. Lindner HH, Kemprud E. A clinicoanatomical study of the arcuate ligament of the diaphragm. Arch Surg 1971 Nov;103(5):600-605.
- 7. You JS, Cooper M, Nishida S, Matsuda E, Murariu D. Treatment of median arcuate ligament syndrome via traditional and robotic techniques. Hawaii J Med Public Health 2013 Aug;72(8):279-281.
- 8. Sapadin A, Misek R. Atypical presentation of median arcuate ligament syndrome in the emergency department. Clin Pract Cases Emerg Med 2019 Oct;3(4):413-416.
- 9. Khrucharoen U, Juo Y-Y, Sanaiha Y, Finn JP, Jimenez JC, Dutson EP. Factors associated with symptomology of celiac artery compression and outcomes following median arcuate ligament release. Ann Vasc Surg 2020 Jan;62:248-257.
- 10. Ologun GO, Snyder H, Hannigan C, Njoku D, Aman M. Celiac artery compression syndrome in a middle-age woman treated laparoscopically. Cureus 2019 Sep;11(9):e5582.
- 11. Torres OJ, Gama-Filho OP, Torres CC, Medeiros RM, Oliveira CM. Laparoscopic treatment of Dunbar syndrome: a case report. Int J Surg Case Rep 2017;37:230-232.