Strongyloides stercoralis is a soil-transmitted intestinal nematode. Its clinical spectrum varies from subclinical to life-threatening disseminated infection. Endogenous autoinfection is a unique feature in strongyloidiasis that may result in a long-lasting infection.1 In the immunocompetent host, cell-mediated immunity plays a role in regulating the population density of adult worms in the intestine. Once the immune system gets compromised, even one adult female can multiply rapidly leading to accelerated autoinfection and/or dissemination.2 Administration of corticosteroids regardless of dose, duration, or route has been described as a risk factor for strongyloidiasis.3 Dexamethasone therapy, due to its powerful anti-inflammatory effects, has been widely used for the management of hospitalized patients with severe COVID-19 as it showed a survival benefit.4 In Oman, the prevalence rate of S. stercoralis is 3% according to studies done in 2013.5 We reported a case of strongyloidiasis with cholestasis post a course of steroid therapy for the management of severe COVID-19 infection.
Case report
A 55-year-old Omani man who works as administrative in the Ministry of Education, presented in May 2021 with a three-week history of profuse diarrhea associated with mucus and a frequency of 10 times per day. The patient had nausea, poor oral intake, and weight loss of approximately 10 kg. He had no history of fever, skin rash, jaundice, or respiratory symptoms. There was no history of travel, change in dietary habits, or ingestion of unusual food, and none of his family members had similar symptoms. His medical background was only remarkable for COVID-19 pneumonia in April 2020 for which he required hospital admission for seven days. During his admission, he received a five-day course of dexamethasone 6 mg/day and was discharged on a prolonged course of tapering dexamethasone regime of 10 mg for five weeks.
The patient was initially evaluated at a local primary health care facility and suspected to have pinworms (Enterobius vermicularis) for which he was treated with a single dose of albendazole 400 mg to be followed by a second dose after two weeks, but the second dose was not taken. His symptoms persisted, therefore he presented to the Royal Hospital for further management. On presentation, the laboratory workup showed high eosinophilia of 1.2 × 109/L (normal range = 0.1–0.5 × 109/L), a picture of cholestasis with alkaline phosphatase levels of 217 IU/L (normal range = 46–116 IU/L), alanine transaminase levels of 76 IU/L (normal range = 10–49), and normal bilirubin 6 µmol/L (normal range = 5–21). Stool microscopy showed Strongyloides larvae in three stool samples [Figure 1]. The abdominal ultrasound showed common bile duct dilation [Figure 2]. Magnetic resonance cholangiopancreatography showed no abnormalities.
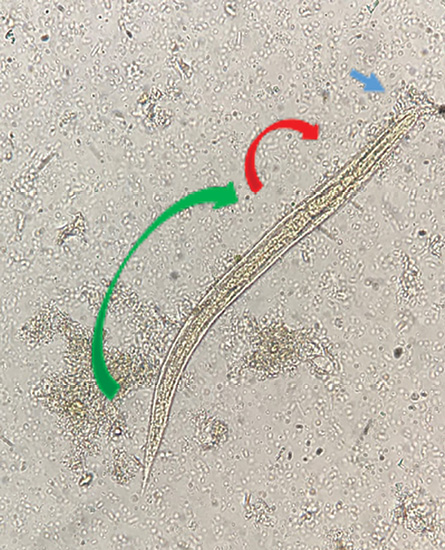 Figure 1: Rhabditiform larva of Strongyloides stercoralis in iodine stain stool sample. It has a short buccal canal (blue arrow), a bulbar portion of the rhabditoid esophagus with three portions (red arrow), and a prominent genital primordium
Figure 1: Rhabditiform larva of Strongyloides stercoralis in iodine stain stool sample. It has a short buccal canal (blue arrow), a bulbar portion of the rhabditoid esophagus with three portions (red arrow), and a prominent genital primordium
(green arrow).
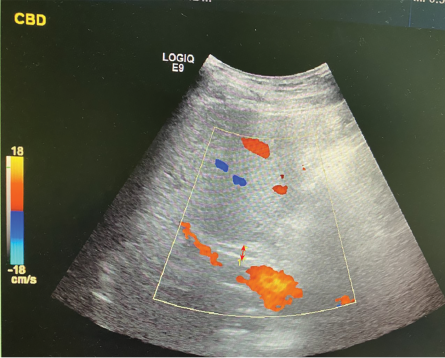 Figure 2: Abdomen ultrasound showed mildly dilated common bile duct.
Figure 2: Abdomen ultrasound showed mildly dilated common bile duct.
The patient was admitted for hydration and was treated initially with two doses of albendazole 400 mg for two days due to the inavailability of ivermectin. He clinically improved and was discharged home. One week later, the patient was reviewed in the clinic, and he complained of persistent diarrhea, so he was given ivermectin 12 mg for two days and repeated it after two weeks. After one month of follow-up, the patient reported complete resolution of symptoms and microbiological clearance as a repeated stool sample tested negative for S. stercoralis.
Discussion
In this report, we discuss a case of strongyloidiasis with cholestasis in an immunocompetent man who has been on a prolonged course of steroid therapy for the management of severe COVID-19 infection and recovered well after three doses of albendazole and two courses of ivermectin.
Most Strongyloides infections (51–64%) are asymptomatic.6 Acute infection may give a characteristic cutaneous reaction as the larvae penetrate the skin or respiratory symptoms, such as a dry cough or wheeze as the larva migrates to the lungs, which were not present in our patient. They can stimulate vague gastrointestinal symptoms once the adult worms reach the small intestine, such as diarrhea, vomiting, and epigastric pain, and our patient had these symptoms.6 Hepatobiliary manifestations of strongyloidiasis seem to be rare, the larvae can cause granulomatous hepatitis with or without larval remnants. Steatosis and cholestasis are other hepatobiliary features and our patients had laboratory and radiological features suggestive of cholestasis.6 Diagnosis of strongyloidiasis by microscopic examination of a single stool sample can detect larvae in about 25% of uncomplicated Strongyloides infections.5 However, repeated examination of concentrated stool samples increases the sensitivity; a number of stool samples recommended is three to seven.5 In our patient, the larva was seen in all three stool samples sent. Peripheral eosinophilia may be associated with parasitic infection and is commonly believed to be a good clinical marker for S. stercoralis infection as was seen in our patient. However, eosinophilia has poor sensitivity, specificity, and predictive value especially in predicting hyperinfection syndrome.7
In our patient, the only risk factor for acquiring strongyloidiasis is the use of steroids as part of COVID-19 management. The strongest risk factor for strongyloidiasis seems to be the corticosteroids, even at a dose of oral prednisolone as low as 20 mg per day and as short as six days.8,9 Corticosteroids were found to increase susceptibility to this parasitic infection through their suppressive effects on some of the major mediators of the immune response like eosinophils.10 There are two previous case reports of patients who developed strongyloidiasis after receiving steroids for COVID-19 treatment, one from Italy and one from the USA.8 Similar to our patient, the two cases have severe COVID-19 pneumonia and received prolonged steroids, the first case presented with abdominal pain and itching and improved with ivermectin, but the second case has respiratory symptoms and required the addition of albendazole to ivermectin for two weeks.9 Ivermectin 200 mcg/kg orally once a day for two days has been used for the management of uncomplicated infection and is generally well-tolerated as have been with our patient.11 However, in our patient, we had to repeat another course of ivermectin as he remained symptomatic. His parasitic load was high due to the prolonged course of dexamethasone which was extended to five weeks. In addition, our patient has cholestasis which could explain his requirement for two courses of ivermectin in addition to three doses of albendazole, which also could play a role in his recovery. However, this will be individualized according to clinical response to treatment. In patients who require prolonged use of steroids for conditions such as necrotizing pneumonia, we propose a risk assessment and screening algorithm for strongyloidiasis in COVID-19 patients. Patients deemed at high risk (including travelers or migrants from endemic areas, farmers, military personnel, refugees, barefoot walkers, and contact with human sewage) may need empirical treatment with ivermectin, which has an efficacy of 85% as a single dose.12
Conclusion
In patients with COVID-19 pneumonia, we suggest avoiding prolonged steroid use to mitigate the risk of strongyloidiasis. For those who will still require steroids as part of COVID-19 treatment, a high index of suspicion and epidemiological risk assessment are the cornerstones for the diagnosis. In such patients, a screening test and an empirical treatment for Strongyloides should be considered to have a better outcome.
Disclosure
The authors declared no conflicts of interest. Written consent was obtained from the patient.
references
- 1. Prevention CC for DC and. CDC - Strongyloides - Biology [Internet]. 2019 [cited 2023 Feb 20]. Available from: https://www.cdc.gov/parasites/strongyloides/biology.html.
- 2. Ramanathan R, Nutman T. Strongyloides stercoralis infection in the immunocompromised host. Curr Infect Dis Rep 2008 May;10(2):105-110.
- 3. Ghosh K, Ghosh K. Strongyloides stercoralis septicaemia following steroid therapy for eosinophilia: report of three cases. Trans R Soc Trop Med Hyg 2007 Nov;101(11):1163-1165.
- 4. Horby P, Lim WS, Emberson JR, Mafham M, Bell JL, Linsell L, et al; RECOVERY Collaborative Group. Dexamethasone in hospitalized patients with Covid-19. N Engl J Med 2021 Feb;384(8):693-704.
- 5. Schär F, Trostdorf U, Giardina F, Khieu V, Muth S, Marti H, et al. Strongyloides stercoralis: global distribution and risk factors. PLoS Neglected Tropical Diseases 2013 Jul 11;7(7):e2288.
- 6. Strongyloides stercoralis. ScienceDirect topics. 2020 [cited 2021 Aug 26]. Available from: https://www.sciencedirect.com/topics/biochemistry-genetics-and-molecular-biology/strongyloides-stercoralis.
- 7. Stauffer WM, Alpern JD, Walker PF. COVID-19 and dexamethasone: a potential strategy to avoid steroid-related strongyloides hyperinfection. JAMA 2020 Aug;324(7):623-624.
- 8. Greaves D, Coggle S, Pollard C, Aliyu SH, Moore EM. Strongyloides stercoralis infection. BMJ 2013 Jul;347:f4610.
- 9. Healio. Strongyloides hyperinfection and its association with COVID-19 treatment. 2021 [cited 2021 Aug 22]. Available from: https://www.healio.com/news/infectious-disease/20210317/strongyloides-hyperinfection-and-its-association-with-covid19-treatment.
- 10. Fardet L, Généreau T, Cabane J, Kettaneh A. Severe strongyloidiasis in corticosteroid-treated patients. Clin Microbiol Infect 2006 Oct;12(10):945-947.
- 11. MSD Manual Professional Version. Strongyloidiasis. 2022 [cited 2021 Aug 15]. Available from: https://www.msdmanuals.com/professional/infectious-diseases/nematodes-roundworms/strongyloidiasis.
- 12. De Wilton A, Nabarro LE, Godbole GS, Chiodini PL, Boyd A, Woods K. Risk of strongyloides hyperinfection syndrome when prescribing dexamethasone in severe COVID-19. Travel Med Infect Dis 2021 Mar-Apr;40:101981.