Mature cystic teratoma (MCT), or dermoid cyst, is a benign germ cell tumor composed of tissue derived from at least two of the three germ layers (endoderm, mesoderm, and ectoderm). Most MCTs are reported in women of reproductive and postmenopausal ages and account for 95% of ovarian germ cell tumors and 20% of all ovarian tumors. Only 10% of MCTs are bilateral.1,2
Malignant transformation of MCT (MT-MCT) can occur in 1–3% of the cases.3,4 The most common MT-MCT is squamous cell carcinoma, representing up to 85% of all cases.5 Others include adenocarcinoma (7%),5 transitional cell carcinoma, malignant melanoma, and carcinoid tumors.2,4 Primary carcinoid tumors in the ovary usually arise in the context of an MCT. These are rare and represent < 0.1% of all ovarian tumors.6
For both adenocarcinoma and primary carcinoid tumor to arise within the same ovarian MCT is extremely rare, especially in young women. This report details such a case in a teenaged patient.
Case Report
In 2017, an 18-year-old woman presented with a left ovarian cyst. She was a patient of neurofibromatosis type 1 (NF1) with an optic nerve glioma that had been surgically removed at the age of 10. During a follow-up visit, a left ovarian cyst was incidentally found and she was referred to the gynecology service.
We found her asymptomatic with no abdominal pain, urinary symptoms, or abnormal bowel habits. Her menstrual cycles were regular, and she was not on any regular medication. On physical examination, a soft pelvic mass was palpable just below the umbilicus. The mass was mobile and nontender. Her serum tumor markers (cancer antigen 125, alpha-fetoprotein, lactate dehydrogenase, and chorionic gonadotropin) were all within normal limits.
A computed tomography scan and magnetic resonance imaging revealed a large complex cyst arising from the left ovary. The cyst measured 13 × 12 × 9 cm, contained mucin, and had solid intramural projections arising from the left lateral wall, measuring 3 × 2 cm [Figure 1]. The residual ovarian tissue showed multiple physiological cysts. The right ovary and uterus were unremarkable. No metastatic pelvic lymph nodes were detected.
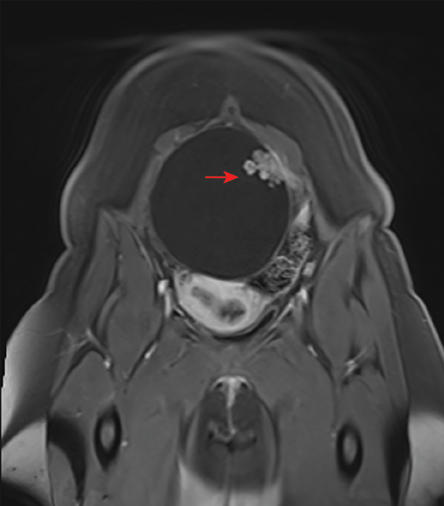 Figure 1: Coronal T2-weighted magnetic resonance image showing left ovarian cystic mass with papillary projections (red arrow).
Figure 1: Coronal T2-weighted magnetic resonance image showing left ovarian cystic mass with papillary projections (red arrow).
The patient underwent laparotomy with left salpingo-oophorectomy, omentectomy, and peritoneal washings. Intraoperatively, the left ovary appeared enlarged, firm, and smooth surfaced—an intact capsule with no surface projections. The right ovary, both fallopian tubes, uterus, bowel, and appendix appeared normal. No enlarged pelvic or para-aortic lymph nodes were noted. There were no ascites or evidence of pelvic disease.
The cyst measured 13 cm in maximum dimension. The ovarian capsule was intact and showed no evidence of surface involvement of the tumor.
When opened, the cyst was found to be filled with yellow mucoid fluid mixed with a cheesy material. The inner surface of the cyst was smooth with a maximum wall thickness of 0.2 cm. There were five solid areas in the form of papillary excrescences, the largest measured 4 cm in greatest dimension. The left fallopian tube was attached to the cyst. The omentum was unremarkable.
The excised sample was sent for histopathological examination. Microscopy showed an MCT with multiple mature elements including squamous epithelium, respiratory epithelium, thyroid tissue, adipose tissue, and cartilage. In addition, an architecturally complex but morphologically bland mucinous proliferation was present with some goblet cells [Figure 2]. Given the architectural complexity, this proliferation was regarded as a grade I mucinous adenocarcinoma arising within a teratoma. A single focus measuring 9 mm in maximum dimension just below the respiratory epithelium, and admixed with bronchial-type epithelial glands showed small cords of bland cells with an epithelioid appearance and abundant clear cytoplasm measuring 9 mm in maximum dimension [Figure 3]. Immunohistochemical staining was performed and these cells were diffusely positive for AE1/AE3, chromogranin, and synaptophysin. There was focal staining with cytokeratin (CK) 7 and occasional cells stained with S100. Transcription termination factor 1, thyroglobulin, inhibin, BerEP4, CK5/6, CD68, CK20, estrogen receptor, human melanoma black 45, prostatic markers, and Napsin A were all negative. There was no nuclear immunoreactivity with β-catenin. Therefore, this focus was considered to represent a proliferation of neuroendocrine cells and regarded as a microscopic carcinoid tumor (grade I neuroendocrine tumor). Both the omentum and peritoneal washings were negative for malignancy. Therefore, the final histopathological diagnosis was grade I mucinous adenocarcinoma with a focus on carcinoid tumor arising within an ovarian MCT.
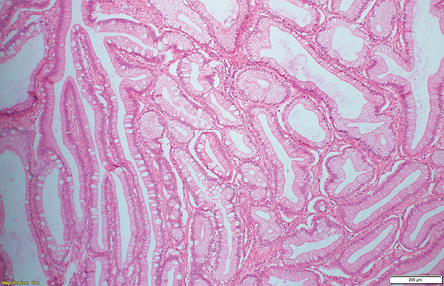 Figure 2: Mucinous tumor arising from the mature teratoma (hematoxylin and eosin stain, magnification = 4 ×). The tumor showed architectural complexity and is composed of bland mucinous proliferation with some goblet cells.
Figure 2: Mucinous tumor arising from the mature teratoma (hematoxylin and eosin stain, magnification = 4 ×). The tumor showed architectural complexity and is composed of bland mucinous proliferation with some goblet cells.
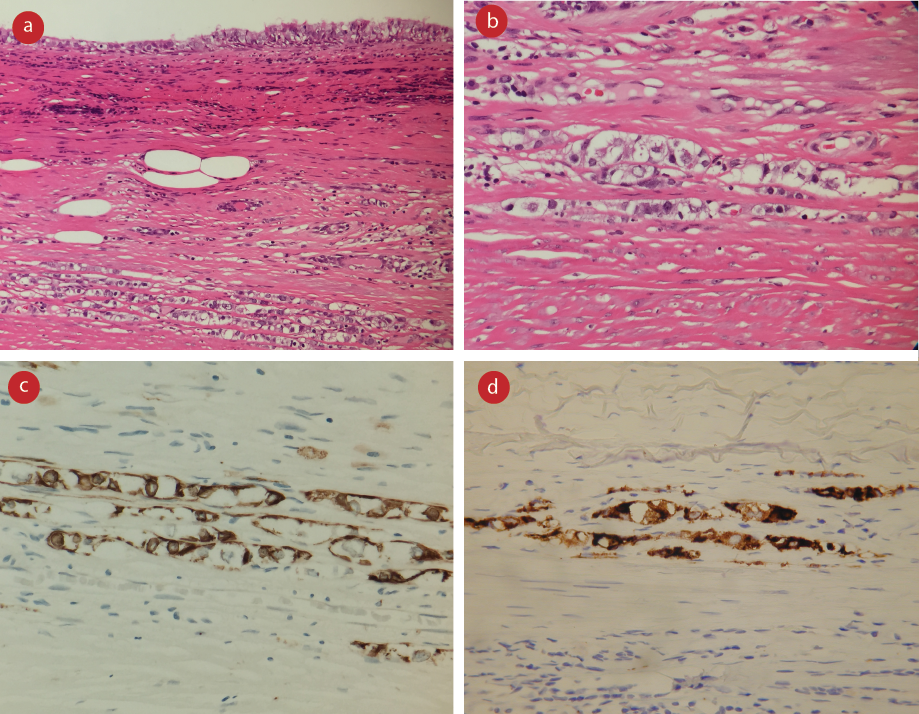 Figure 3: Microscopy of the carcinoid tumor. (a and b) Carcinoid tumor cells arranged as cords below the respiratory epithelium. The cells were bland with epithelioid morphology and abundant clear cytoplasm (hematoxylin and eosin stain, magnification = 40 ×). (c and d) These cells were positive for AE1/AE3 and synaptophysin immunostains, respectively (hematoxylin and eosin stain, magnification = 40 ×).
Figure 3: Microscopy of the carcinoid tumor. (a and b) Carcinoid tumor cells arranged as cords below the respiratory epithelium. The cells were bland with epithelioid morphology and abundant clear cytoplasm (hematoxylin and eosin stain, magnification = 40 ×). (c and d) These cells were positive for AE1/AE3 and synaptophysin immunostains, respectively (hematoxylin and eosin stain, magnification = 40 ×).
The hospital’s tumor board deemed the treatment complete based on the presence of the intact capsule and the early stage of the disease. Surveillance and genetic counseling were recommended at this point, considering the patient’s history of NF1. She has been regularly followed-up since with no evidence of recurrence or metastasis.
Discussion
In this case report, we have demonstrated dual tumors—a mucinous adenocarcinoma and a carcinoid tumor—arising within the same ovarian MCT in an 18-year-old woman. What makes this case rare and unique are the youth of the patient, and the extremely rare occurrence of two different types of malignancies within the same MCT.
MT-MCT is most frequently seen in older, especially menopausal, women, with a mean age of incidence of 51.3 years. Even for pure (benign) MCT, the mean age is 45.3 years. In one observational study, nine out of 21 postmenopausal women with MCT had a MT.7 The pathogenesis of this transformation is hypothesized to stem from a long-standing unresected benign teratoma. Some small-scale molecular studies have also shown evidence of p53 overexpression, particularly in cases with squamous cell carcinoma.4,8
Clinically, the most common presenting complaints in women with MT-MCT are abdominal pain, pelvic pain, and abdominal mass. Occasionally, patients are asymptomatic, and the mass is detected incidentally, as in the present case.7 While MCT can be diagnosed radiologically, MT-MCT cannot be readily distinguished by imaging alone.4,7 The diagnosis of MT-MCT is usually made postoperatively by histopathology.
There are 42 documented cases in the literature of adenocarcinoma arising from an MCT, and 33% of these cases are of mucinous adenocarcinoma.5 Primary carcinoid tumors of the ovary are rare and account for only 0.3% of all carcinoid tumors, and < 0.1% of all ovarian tumors. Carcinoid tumors arising in an ovarian MCT are more common than primary carcinoid tumors.9 They originate from neuroendocrine cells of mature epithelium, and are described as having low malignant potential.6,10 In the present case, it was a grade I neuroendocrine tumor arising just beneath the respiratory epithelium.
There is a link between our young patient’s chronic NF1 and the different neoplasms she presented with. NF1 is an autosomal dominant disorder caused by mutations in the NF1 tumor suppressor gene. It is commonly associated with neurogenic and central nervous system neoplasms in addition to a wide variety of other benign and malignant tumors.11,12 The possibility of teratomas arising secondary to biallelic inactivation of the NF1 gene has been suggested, though based on a single case.11 Gastrointestinal tract (GIT) neoplasms have also been reported.12 The most common GIT manifestation is gastrointestinal stromal tumor, which lacks the characteristic mutations commonly associated with it.13 Adenocarcinoma has been reported along with neuroendocrine tumors of the GIT.12 In fact, most of the neuroendocrine tumors described in patients with NF1 involve the GIT.
A search for published case reports of synchronous two or more malignant tumors in an MCT yielded very few results. One case report describes a colonic adenocarcinoma with a well-differentiated neuroendocrine tumor.14 Another case details a mucinous adenocarcinoma and strumal carcinoid tumor.15 Both patients were premenopausal women.
For the diagnosis of MT-MCT, careful macroscopic examination and adequate sampling of all solid areas in the specimen are essential for accurate identification of all teratoma components. Complete surgical resection remains the mainstay of treatment of MT-MCT and shows better overall survival. Recent evidence suggests that chemotherapy may improve overall survival.16–18
Conclusion
MT-MCT is a rare complication of MCT and is mostly reported among postmenopausal women. It is extremely rare for malignant transformation of more than one component of the MT to occur, especially in young women. This case highlights the importance of thorough gross examination and adequate sampling of all solid and suspicious areas in the ovarian cyst wall to avoid missing any rare or synchronous malignancies.
Disclosure
The authors declared no conflicts of interest. Written consent for publication has been obtained from the patient’s next of kin. Abstract of this paper was published in the 2018 XXXII Congress of the International Academy of Pathology, Jordan.
Acknowledgments
We greatly appreciate the expert advice and review of the case by Professor W. Glenn McCluggage, consultant histopathologist in Royal Group of Hospitals Trust, Belfast, and Honorary Professor in gynecological pathology at Queen’s University of Belfast, Northern Ireland.
references
- 1. Vang R, Zaloudek C. Tumors of the ovary: mature teratoma of the ovary. WHO classification of tumours. 5th ed. International Agency for Research on Cancer (IARC); 2020:119-120.
- 2. Welter SM, Khalifa MA. Ovary. Germ cell tumors, teratoma-mature. Pathologyoutlines.com. 2021 [cited 2022 January 18]. Available from: https://pathologyoutlines.com/topic/ovarytumorteratomamature.html.
- 3. Tehranian A, Ghahghaei-Nezamabadi A, Seifollahi A, Kasraei S, Dehghani-Nejad H, Maleki-Hajiagha A. Ovarian mature cystic teratoma with malignant transformation: two case reports. J Med Case Rep 2021 Jan;15(1):23.
- 4. Rathore R, Sharma S, Agarwal S. Malignant transformation in mature cystic teratoma of the ovary: a retrospective study of eight cases and review of literature. Menopause Review/Przegląd Menopauzalny 2018;17(2):63-68.
- 5. Miyasaka A, Nishikawa T, Kozawa E, Yasuda M, Fujiwara K, Hasegawa K. Advanced mucinous adenocarcinoma arising from a mature cystic teratoma: a case report and literature review. Case Rep Oncol 2016 Jun;9(2):331-337.
- 6. Kim JY. A carcinoid tumor arising from a mature cystic teratoma in a 25-year-old patient: a case study. World J Surg Oncol 2016 Apr;14:120.
- 7. Qin L, Zhao T, Liu X, Wang H, Gu X, Chen D, et al. Malignant transformation arising from mature ovarian cystic teratoma: a case series. Medicine (Baltimore) 2021 Apr;100(13):e24726.
- 8. Yoshioka T, Tanaka T. Immunohistochemical and molecular studies on malignant transformation in mature cystic teratoma of the ovary. J Obstet Gynaecol Res 1998 Apr;24(2):83-90.
- 9. Baker P, Rabban J, Matias-Guiu X. Tumors of the ovary: ovarian carcinoid. WHO classification of tumours. 5th ed. International Agency for Research on Cancer (IARC);2020:134-135.
- 10. Tosuner Z, Sönmez FC, Arici DS, Dansuk R. Carcinoid tumor arising in a mature cystic teratoma: a case report. Oncol Lett 2015 May;9(5):2236-2238.
- 11. Yap P, Super L, Qin J, Burgess T, Prodanovic Z, Edwards C, et al. Congenital retroperitoneal teratoma in neurofibromatosis type 1. Pediatr Blood Cancer 2016 Apr;63(4):706-708.
- 12. Abdessayed N, Gupta R, Mestiri S, Bdioui A, Trimech M, Mokni M. Rare triad of periampullary carcinoid, duodenal gastrointestinal stromal tumor and plexiform neurofibroma at hepatic hilum in neurofibromatosis type 1: a case report. BMC Cancer 2017 Aug;17(1):579.
- 13. Garrouche N, Ben Abdallah A, Arifa N, Hasni I, Ben Cheikh Y, Ben Farhat W, et al. Spectrum of gastrointestinal lesions of neurofibromatosis type 1: a pictorial review. Insights Imaging 2018 Oct;9(5):661-671.
- 14. Ayyanar P, Begum J, Rout S, Mishra P. Synchronous colonic adenocarcinoma and well-differentiated neuroendocrine tumor arising in a mature cystic teratoma of ovary - rare presentation in a postmenopausal woman with literature review. Indian J Pathol Microbiol 2021;64(2):385-389.
- 15. Kim SM, Choi HS, Byun JS, Kim YH, Kim KS, Rim SY, et al. Mucinous adenocarcinoma and strumal carcinoid tumor arising in one mature cystic teratoma of the ovary with synchronous cervical cancer. J Obstet Gynaecol Res 2003 Feb;29(1):28-32.
- 16. Mandal S, Badhe BA. Malignant transformation in a mature teratoma with metastatic deposits in the omentum: a case report. Case Reports in Pathology 2012;2012:568062.
- 17. Wang J, Kazmi SA. Teratoma with malignant transformation: a case report with pathological, cytogenetic, and immunohistochemistry analysis. Sarcoma 2011;2011 :450743.
- 18. Donadio AC, Motzer RJ, Bajorin DF, Kantoff PW, Sheinfeld J, Houldsworth J, et al. Chemotherapy for teratoma with malignant transformation. J Clin Oncol 2003 Dec;21(23):4285-4291.