Cyclosporine A (CsA) is a known immunosuppressant that is widely used in multiple medical specialties, such as in the treatment of multiple transplants and skin and rheumatology conditions.1 CsA is a calcineurin inhibitor that inhibits cell-mediated and humoral immune responses by binding to cyclophilin, leading to the activation of nuclear factor of activated T cells and reduction in interleukin-2 transcription. The known side effects of this medication are renal toxicity, hypertension, hypertriglyceridemia, immunosuppression, and lymphoma.2 CsA can affect hair by causing hypertrichosis, though it is rarely reported to cause hair darkening.3
We report a case of an Omani male who presented to the dermatology clinic at Rustaq Polyclinic in Rustaq, Oman in 2020, with a reversal of his hair color from white to black after three months of treatment with CsA for chronic atopic dermatitis.
Case report
A 65-year-old Omani male, who was a known smoker with no comorbid conditions and not on any medications, attended the dermatology clinic for chronic atopic dermatitis for 11 years. He first presented with itchy scaly erythematous plaques over exposed areas, which were initially controlled by topical steroids for several years. For the last three years, he presented with multiple flare-ups, which were controlled with topical steroids, tapering oral steroid courses, and antihistamines.
In mid-2020, he developed generalized exfoliative erythroderma, which required hospital admission. A skin biopsy was done, which indicated the presence of spongiotic dermatitis and no evidence of malignancy. He underwent pan-computed tomography, which showed no evidence of internal malignancy. Blood tumor markers (including alpha-fetoprotein, carcinoembryonic antigen, and prostate-specific antigen) were normal. His peripheral smear was reviewed by a hematologist and was normal.
He was started on methotrexate (MTX), 7.5 mg once a week titrated slowly to 20 mg. Despite being on a high dose of MTX, the patient’s condition was worsening regarding his dermatitis and itching. The MTX was stopped and was changed to CsA within a month, initially 3 mg/kg/day divided into two doses for two weeks then titrated to 5 mg/kg/day. The patient showed improvement using CsA with no major side effects noticed on follow-ups.
After three months of treatment, he developed hypertrichosis and reversal of his hair color from white [Figure 1] to black [Figure 2], which first appeared over scalp edges, central scalp, beard, and moustache, then slowly spread to the other scalp areas.
 Figure 1: The patient one year before starting cyclosporine A.
Figure 1: The patient one year before starting cyclosporine A.
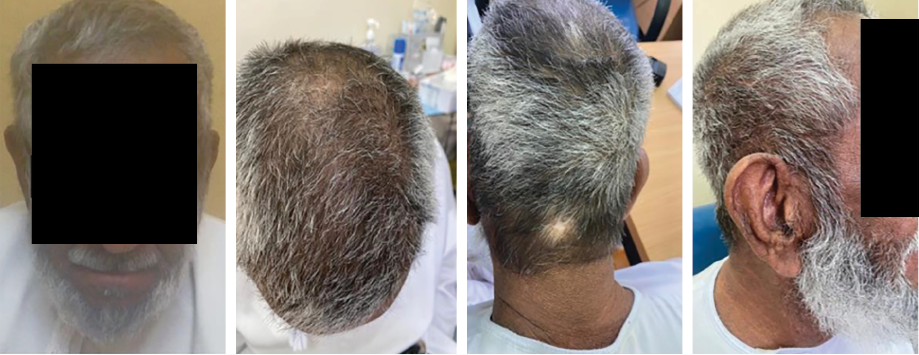 Figure 2: The patient three months after starting cyclosporine A, showed darkening of hair over the scalp, moustache, and beard.
Figure 2: The patient three months after starting cyclosporine A, showed darkening of hair over the scalp, moustache, and beard.
Discussion
Diffuse repigmentation of gray hair can be induced by certain medications that inhibit inflammation or stimulate melanogenesis. Many drugs have been reported to cause hair darkening, including anti-inflammatory medications.4
CsA is known to cause dose-dependent hypertrichosis, which has been reported in approximately 50% of transplant patients who take a high dose of the drug, and in around 3% of patients with skin conditions, especially those who have been treated with 5 mg/kg/day of CsA.5
CsA causes hypertrichosis by inducing and prolonging the anagen growth phase, which enhances hair follicle stem cell activity, and blocks the ability of the dermal papilla cells to initiate catagen.6 Through interfering with T cell activity and regulation of interleukin-2 activity, CsA plays a role in the activation of degenerated or dormant melanocyte stem cells and recruits functional melanocyte stem cells from neighboring hairs. One randomized control study on mice involving a new compound (CsA, minoxidil, and a pigment-promoting drug) induced gray hair repigmentation. The increase in the pigmentation of gray mouse hairs was associated with increased melanocyte progenitor cell count in up to 80% of the hair bulbs. The study also showed that even after stopping the treatment, shaved hair was noted to regrow with continued repigmentation in following hair cycles.7
Only two cases of hair darkening after introduction of CsA have been reported in the literature.8,9 One was in a 59-year-old man with psoriasis who presented with erythroderma and had been on MTX for 20 years. MTX was stopped because the patient developed liver cirrhosis and he was switched to 5 mg/kg/day of CsA. Hypertrichosis and hair darkening occurred after two months of treatment, where it first appeared on the scalp edges.8 The second case was a 73-year-old man with eczematous dermatitis, who developed erythroderma and was treated with 5 mg/kg/day CsA. Hair darkening was noticed one month after treatment.9
The patient in this case and the other two previously reported cases shared similar backgrounds of autoinflammatory dermatological conditions progressing to erythroderma, requiring the initiation of CsA. In addition, all three patients developed hair darkening on the same dose of CsA, 5 mg/kg/day, and over similar time periods.
Conclusion
CsA causes hypertrichosis and repigmentation of the hair, beginning as little as a month after treatment. To know whether this side effect is temporary or permanent, more case reports are needed with prolonged follow-up for the patients after stopping CsA. Despite some proposed mechanisms explaining the hair repigmentation, further studies are needed to determine the exact mechanism.
Disclosure
The authors declared no conflicts of interest. Informed written consent was taken from the patient .
references
- 1. Smith SG. Neoral (cyclosporin) in dermatology: technical aspects. Br J Dermatol 1996 Sep;135(48)(Suppl 48):2-4.
- 2. Bolognia JL, Schaffer JV, Cerroni L. Dermatology. 4th ed. Philadelphia: Elsevier; 2018. p. 2288.
- 3. Ricci F, De Simone C, Del Regno L, Peris K. Drug-induced hair colour changes. Eur J Dermatol 2016 Dec;26(6):531-536.
- 4. Yale K, Juhasz M, Atanaskova Mesinkovska N. Medication-induced repigmentation of gray hair: a systematic review. Skin Appendage Disord 2020 Jan;6(1):1-10.
- 5. Tosti A, Pazzaglia M. Drug reactions affecting hair: diagnosis. Dermatol Clin 2007 Apr;25(2):223-231.
- 6. Hawkshaw NJ, Haslam IS, Ansell DM, Shamalak A, Paus R. Re-evaluating cyclosporine A as a hair growth–promoting agent in human scalp hair follicles. J Invest Dermatol 2015 Aug;135(8):2129-2132.
- 7. Anderson ZT, Palmer JW, Idris MI, Villavicencio KM, Le G, Cowart J, et al. Topical RT1640 treatment effectively reverses gray hair and stem cell loss in a mouse model of radiation-induced canities. Pigment Cell Melanoma Res 2021 Jan;34(1):89-100.
- 8. Sadighha A, Zahed GM. Hair darkening after treatment with cyclosporin in a patient with psoriasis. J Eur Acad Dermatol Venereol 2008 Nov;22(10):1239-1241.
- 9. Rebora A, Delmonte S, Parodi A. Cyclosporin A-induced hair darkening. Int J Dermatol 1999 Mar;38(3):229-230.