Disseminated tuberculosis (TB) results from massive lymphohematogenous dissemination of Mycobacterium tuberculosis. Clinical manifestations of disseminated TB are most likely to be subacute or chronic. Less commonly, acute presentations also occur.1,2 Patients with subacute or chronic TB may present with fever of unknown origin, night sweats, fever, rigor, and/or dysfunction of one or more organs. Acute presentation, including multisystem failure, septic shock, and acute respiratory distress syndrome.3 Severe hypokalemia as a presenting feature of disseminated or miliary TB is a rare entity, and the association between disseminated TB and hypokalemia is poorly understood. To highlight this rare association and to explain the possible association between these two clinical conditions, we report a case of a 38-year-old male patient who presented with weakness. He had severe hypokalemia, and further investigations revealed disseminated TB.
Case report
A previously healthy, 38-year-old male presented with generalized body weakness for five days. Described as general weakness more in the lower limbs associated with generalized fatigability, he denied any muscle pain, specific proximal or distal distribution, or any variation during the day. Over the next five days, his weakness was progressively increasing. He had difficulty walking, leading to a fall. This weakness was associated with on and off fever, occasional dry cough, and two episodes of vomiting. The patient had polyuria, polydipsia, and nocturia for two days, which developed for the first time.
Patient denied having headache, photophobia, phonophobia, neck pain or stiffness, abnormal jerky movements, palpitations, sweating, weight loss, shortness of breath, sputum production or dysuria, and no history of diarrhea or poor dietary intake. The patient did not smoke, drink alcohol, or use illicit drugs. He was not sexually active. He worked as a salesman and denied sick contacts or recent travel.
Upon presentation to the emergency department, the patient was febrile, but other vitals were stable. He needed support to walk to the bathroom. Upon examination, his lower limbs power was 4/5, which temporarily improved to 5/5 after intravenous (IV) potassium infusion. Meningeal signs were negative, and the rest of his physical exam was unremarkable. Initial labs showed severe hypokalemia of 2.1 mEq/L (normal 3.5–5 mEq/L) and hyponatremia of 127 mEq/L (normal 135–145 mEq/L), creatinine 73 umol/L (normal 62–106 umol/L), chloride 83 mmol/L (normal 90–105 mmol/L), bicarbonate 29 mmol/L (normal 22–29 mmol/L), corrected calcium 2.54 mmol/L (normal 2.2–2.55 mmol/L), glucose 7.9 mmol/L (3.5–5.5 mmol), repeated random glucose readings were normal, glycated hemoglobin (HB1c) 5.6% (normal < 6.5%), lactic acid 2.0 (normal < 2), hemoglobin 13.5 gm/dL (normal 13–17 gm/dL), white blood cells (WBC) 19.10 × 103/uL (normal 4–10 × 103/uL), HIV was negative, and urine dipstick was unremarkable. Electrocardiogram showed regular sinus rhythm, T wave inversion in inferolateral leads, U wave, and epsilon wave. The patient was admitted as a case of severe symptomatic hypokalemia and fever for workup and management.
His 24 hour urine potassium after 48 hours admission was 156 mmol/24 hours in 4.6 L of urine (normal 25–125 mmol/24 hour). IV potassium replacement was started. The patient received a total of 110 mEq IV and 60 mEq oral in the first 24 hours of his hospital stay. His potassium remained low at 2.5 mEq/L (normal 3.5–5 mEq/L). The patient was kept on continuous IV potassium replacement (4 mmol/hr) in addition to 30 mEq oral q8hrs over the next two days. His potassium improved to 3.9 mEq/L, and his weakness disappeared completely.
Sepsis screen came unremarkable, including two sets of blood cultures, urine culture, chest X-ray (CXR), and abdominal ultrasound. The patient was started on ceftriaxone q24hr initially, but after 48 hours hospital stay, he still had a fever, and reported new neck pain. Upon examination, he was found to have neck stiffness, positive Kernig’s and Brudzinski signs, new bilateral lower limb weakness (power 3/5), and urine retention. Lumbar puncture (LP) showed cerebrospinal fluid (CSF) protein 5.7 gm/L (normal 0.15–0.45 gm/L), CSF glucose 0.97 mmol/L (normal 2.2–
3.8 mmol/L), CSF WBC 484/uL (normal 0–5/uL), 78% lymphocytes, and 16% neutrophils. Ceftriaxone changed to q12hr and started on anti-TB treatment with prednisolone 60 mg and pyridoxine, then CSF TB polymerase chain reaction (PCR) came positive, so ceftriaxone was discontinued, and he was continued on anti-TB treatment. Sputum for TB acid-fast bacilli smear, PCR, and culture were induced with hypertonic saline and sent. Repeated LPs after three days showed improvement with CSF protein decreased to 1.22 gm/L. CSF glucose increased to 1.62 mmol/L. Magnetic resonance imaging of the brain showed features highly suggestive of TB meningitis involving the brain and spinal canal with the small right frontal and left cerebellar tuberculoma, respectively. We suspected mild supratentorial hydrocephalus, but the images were reviewed and cleared by the radiologist.
The patient’s condition improved, and he became afebrile but with remnant lower limb weakness and urine retention. He was transferred to rehabilitation medicine on day 17 of admission. His potassium levels improved gradually and normalized after 12 days then supplements were stopped on day 13 of hospital stay [Figure 1]. Four weeks from admission, his sputum TB culture came back positive (rifampicin and isoniazid (INH) sensitive) and patient was labeled as disseminated TB. The patient continued treatment in a rehabilitation hospital for five weeks and was then discharged on anti-TB treatment with direct observation therapy and infectious disease physician follow-up. His potassium remained normal in the rehabilitation period and did not require any supplements.
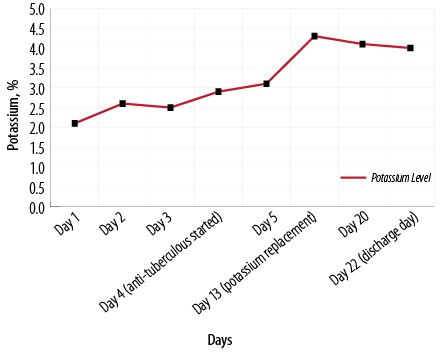
Figure 1: Correlation between potassium level and treatment of the patient.
Discussion
Disseminated TB results from massive lymphohematogenous dissemination of Mycobacterium tuberculosis. Electrolytes disturbances could be associated with TB. Hyponatremia is frequently observed and reported; it is presumed to be due to dysregulation of antidiuretic hormone (SIADH), causing more water retention with excessive sodium excretion from the urine.4
Hypokalemia is one of the adverse reactions commonly found in patients with multi-drug resistant TB (MDR-TB) treated with tablets or injectable agents. Rifampicin, amikacin, and viomycin-pyrazinamide are thought to be associated with electrolytes imbalance, including hypokalemia.5–8
Hypokalemic periodic paralysis is also considered one of the well-known causes of hypokalemia which has an autosomal dominant mode of inheritance, or it can be acquired in patients with hyperthyroidism. Acute attacks which cause sudden movement of potassium into the cells are often precipitated by rest after exercise, stress, or a carbohydrate meal. In our patient, infection with TB could be the stressful trigger causing hypokalemia.
Hypokalemia is a rare association with pulmonary TB. One case report mentioned the association of Bartter syndrome with pulmonary TB in a middle-age lady who presented with polyuria and frequency, wasting syndrome, and cramps. Laboratory evaluation showed normal leukocytosis (12.100/mm3), hyponatremia (123 mEq/L), severe hypokalemia (1.34 mEq/L), blood urea nitrogen (67 mg/dL), metabolic alkalosis pH 7.40, HCO3¯= 30 mol/L. A 24-hour urine collection (3500 mL) revealed a potassium level of 40 mEq/L. The patient was treated with regular therapy with oral potassium 15 g/day for almost five years without significant improvement in her symptoms. The patient was admitted again with fever, chills, cough, wasting syndrome, polyuria, and polydipsia and found to have severe hypokalemia. Further evaluation, including CXR, showed opacity in her upper left pulmonary lobe and pulmonary TB was also confirmed by positive smears and cultures. The authors suggested that tubulopathies caused by primary infection explained the unusual association between Bartter syndrome and pulmonary TB.9
Hypomagnesemia contributing to hypocalcemia and hypokalemia, occurring as a primary abnormality in patients with pulmonary TB was reported by Baskaran et al,10 which was worsened by the use of streptomycin. The authors mentioned that hypomagnesemia is multifactorial in origin, including malnutrition, malabsorption, and therapy-induced renal loss.
To the best of our knowledge, this is the first case report on the association between disseminated TB and severe hypokalemia. We reemphasized the assumption that TB, which is in our case the disseminated type, can lead to tubular defect, which can cause potassium losing nephropathy. Future studies may highlight the exact mechanism.
Conclusion
Despite severe hypokalemia being a rare association with TB, disseminated TB can be considered the possible cause in patients presenting with severe hypokalemia and fever. Renal tubular defect could be the proposed mechanism causing potassium losing nephropathy.
Disclosure
The authors declared no conflicts of interest. Informed patient consent has been taken before reporting this case.
references
- 1. Ellner JJ. Tuberculosis. In: Goldman L, Schafer AI, editors. Goldman-cecil medicine. 25th ed. Philadelphia, PA: Elsevier Saunders; 2016.
- 2. Khan FY, Dosa K, Fuad A, Ibrahim W, Alaini A, Osman L. Disseminated tuberculosis among adult patients admitted to Hamad general hospital, Qatar: a five year hospital based study. Mycobact Dis 2016;6(212):2161-1068.
- 3. Sydow M, Schauer A, Crozier TA, Burchardi H. Multiple organ failure in generalized disseminated tuberculosis. Respir Med 1992 Nov;86(6):517-519.
- 4. Shalhoub RJ, Antoniou LD. The mechanism of hyponatremia in pulmonary tuberculosis. Annals of Internal Medicine 1969 May 1;70(5):943-962.
- 5. Shin S, Furin J, Alcántara F, Hyson A, Joseph K, Sánchez E, et al. Hypokalemia among patients receiving treatment for multidrug-resistant tuberculosis. Chest 2004 Mar 1;125(3):974-980.
- 6. Cheng JT, Kahn T. Potassium wasting and other renal tubular defects with rifampin nephrotoxicity. American Journal of Nephrology 1984;4(6):379-382.
- 7. Faheem JA, Hassan M, Muhamma N, Wali S, Akbar HS, Atta QM, et al. Electrolytes imbalance caused by amikacin in patients receiving multi drug resistance-tuberculosis treatment at Hazara region Kpk, Pakistan. Tuberk Toraks 2017;65(3):193-201.
- 8. Vanasin B, Colmer M, Davis PJ. Hypocalcemia, hypomagnesemia and hypokalemia during chemotherapy of pulmonary tuberculosis. Chest 1972 May 1;61(5):496-499.
- 9. Bontas E, Dumitrescu AM. Bartter’s syndrome with pulmonary tuberculosis: a shortly outlook. Mædica a Journal of Clinical Medicine 2007;2(4):332.
- 10. Baskaran S, Manigandan G, Aashish A. Severe hypomagnesemia, hypokalemia and hypocalcemia associated with pulmonary tuberculosis. International Journal of Current Research and Review 2014 Mar 15;6(6):80.