Splenic abscess has varied etiologies with splenic tuberculosis (TB) being a rare but important presentation of abdominal TB.1,2 Splenic TB is almost always a part of miliary TB, but on rare occasions, isolated splenic involvement or contiguous spread from an abdominal lymph node can occur. Isolated splenic TB is known to occur mainly in HIV-positive or immunocompromised individuals but rarely can occur in immunocompetent patients in endemic areas.3,4 Thus, any patient with non-specific symptoms, fever, abdominal pain, splenomegaly, and splenic abscesses should be evaluated for tuberculosis. Early initiation of anti-tubercular therapy (ATT) and immediate percutaneous drainage procedures can avoid untoward splenectomy and mortality.5
Case Report
A 30-year-old male, a manual laborer by occupation, presented with dragging pain in the left upper quadrant of his abdomen, which lasted for three months. He had a history of progressive loss of appetite and weight (15%). He had no history of fever, previously known comorbidity, and no history of contact to TB. On examination, the patient had normal vital parameters, his general physical examination was unremarkable, and he had no palpable lymph nodes. Abdominal examination revealed an enlarged, non-tender spleen with the lower border palpable 5 cm below the left costal margin. Cardiovascular, respiratory, and central nervous system examination were normal.
Complete blood counts, erythrocyte sedimentation rate (ESR), liver function, renal function, and chest X-ray were within normal limits. Ultrasonography (USG) confirmed splenomegaly with multiple hypoechoic areas with anechoic content, suggestive of multiple abscesses and the largest was 7 × 6.5 cm at the lower pole of the spleen. Subsequently computed tomography (CT) of the abdomen confirmed the same finding and in addition demonstrated multiple calcified foci in the spleen along with few calcified mesenteric and retroperitoneal lymph nodes [Figure 1]. CT chest was normal, and there was no evidence of abscess in any other part of the body.
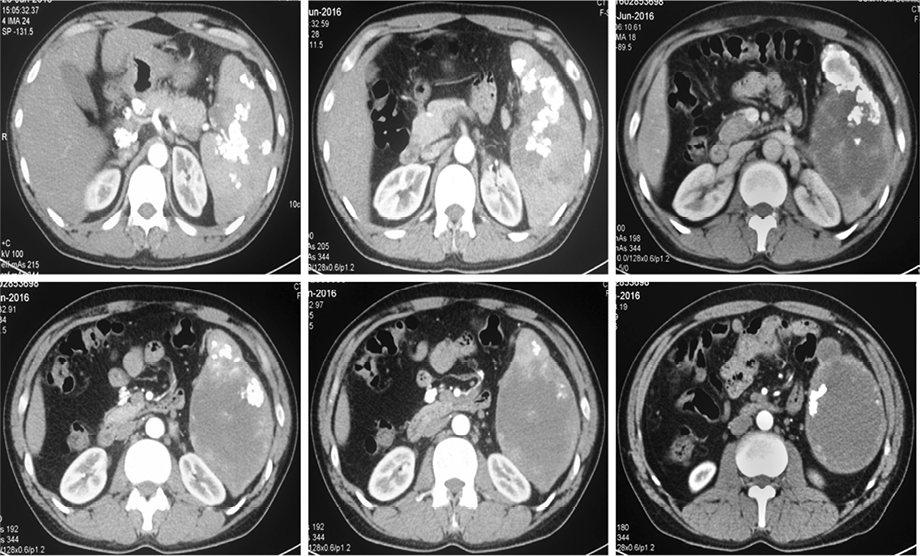
Figure 1: Computed tomography of the abdomen showing enlarged spleen (17.2 cm), multiple irregular splenic hypodense lesions, largest at the lower pole (10 × 5 cm, 25 Hounsfield units), along with multiple hyperdense foci suggesting abscesses with calcification, and few calcified retroperitoneal lymph nodes.
TB, infective endocarditis (IE), malignancy, lymphoma, sarcoidosis, and histoplasmosis were kept among the differential diagnosis. Blood cultures were sterile, transthoracic and transesophageal echocardiography did not reveal any evidence of IE. Mantoux skin test was strongly positive (25 mm). Levels of serum calcium, vitamin D, and parathyroid hormone were normal, and histoplasma urine antigen was negative. Viral markers for HIV, hepatitis B, and hepatitis C were negative. CD4 count, serum immunoglobulin level, and glycated hemoglobin (HbA1c) were within normal range. The Xpert MTB (GeneXpert) test and microbiology smears of the fine-needle aspirate from the spleen confirmed Mycobacterium tuberculosis (MTB) [Figure 2]. Ultrasound-guided percutaneous pigtail drainage of the large collection in the inferior pole of the spleen was done, and the patient was started on four drugs ATT: isoniazid, rifampin, pyrazinamide, and ethambutol. Pneumococcal vaccination was given for the possible need for splenectomy.
Subsequently, there was a gradual decrease in the size of the collection, and the pigtail catheter was removed after seven days when there was no drainable collection. Significant improvement of the constitutional symptoms was noted after
two weeks, and ATT was planned to continue for six months.
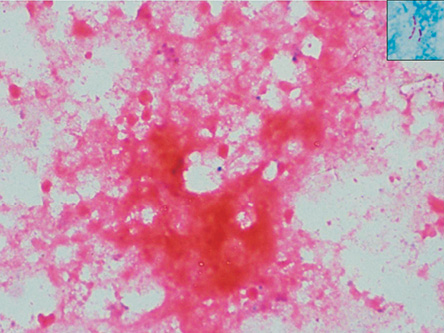
Figure 2: Microphotograph showing caseous necrosis (hematoxylin and eosin stain, magnification = 40 ×) with acid-fast bacilli (inset: Ziehl–Neelsen stain, magnification = 100 ×).
Discussion
Splenic abscess is an extremely uncommon condition with an incidence of 0.14–0.7% as per autopsy studies from western countries.6 However, with improved imaging techniques and increases in the population of patients with immunosuppression and malignancy, an apparent rise in the burden of splenic abscess has been noted.7
The etiology of splenic abscesses may be diverse. The most common cause being hematogenous dissemination to the spleen from an infective focus. IE accounts for 10–20% of all cases.5 The other infective causes include enteric fever, urinary tract infection, pneumonias, osteomyelitis, mastoiditis, tuberculosis, brucellosis, and pelvic infections.3 The aerobic microbes notably Streptococci and Escherichia coli are the organisms reported most frequently from the case series.8 Multiple splenic lesions have also been noted with Salmonella typhi infection and in contrast to prior reports, many studies now reported Klebsiella pneumoniae from splenic abscess aspirates.9 Superinfection of a damaged or infarcted spleen in hemoglobinopathies, leukemias, polycythemia, and penetrating or non-penetrating trauma can also result in abscess formation. Alcoholics, diabetics, and immunosuppressed patients remain at high risk of splenic abscess formation6 and extension from a contiguous focus of infection (e.g., pancreatic, subphrenic, and perinephric areas) can also cause suppurative splenic lesions.
Extrapulmonary TB constitutes 15% of all cases of TB in immunocompetent individuals,10 and abdominal TB constitutes 3–4% of the extrapulmonary burden. Sites of involvement can include most commonly lymph nodes, gastrointestinal tract, peritoneum, and rarely the tongue, esophagus, liver, spleen, and pancreas.11–13 Splenic TB is usually a part of miliary or disseminated TB and rarely may occur in isolation. The spleen is the third most common organ involved after the lung and liver in miliary TB. According to an Indian study on splenic TB, 62% was associated with pulmonary TB, and 50% of patients had HIV coinfection.14 An unusual form of splenic TB is the primary involvement of the spleen appearing as solitary or multiple abscesses. Such isolated foci in the spleen have been reported more frequently in HIV-positive and immunocompromised individuals. However, the index case was immunocompetent and HIV-negative. In another study, 10 cases of splenic TB were reported in immunocompetent patients, although unlike the index case, these patients also showed other organ involvement.3 Singh et al,4 also reported four cases of isolated splenic TB in non-HIV patients, but isolated splenic cold abscesses is still a rare entity. Thus isolated splenic TB should be considered strongly even in HIV-negative individuals from endemic areas who present with fever, constitutional symptoms, and left upper quadrant pain.
Clinical features may be non-specific, and diagnosis of splenic TB is often delayed. Fever, anorexia, weight loss, and abdominal pain should provide a clue, especially in the presence of positive contact history or HIV-positive status. USG and CT play a major role in diagnosis and patient management.4 It helps to assess the nature of the lesion, identify a suitable site for aspiration and in percutaneous drainage. The differential diagnoses of hypodense lesions on abdominal CT are various congenital, parasitic, or neoplastic cysts; abscesses, traumatic, sickle cell disease, infarction, hamartoma, sarcoidosis, hemangiomas, lymphomas, and metastatic deposits.15 Confirmatory diagnosis is obtained only after histopathologic examination of a splenectomy specimen or acid-fast bacilli (AFB)smear/culture of splenic abscess aspirates. The index patient was subjected to guided fine-needle aspiration from one of the splenic lesions followed by pigtail drainage. AFB smears and Xpert MTB of the material aspirated were positive thereby confirming the diagnosis. Xpert MTB has a combined sensitivity and specificity of 77.3% and 98.2%, respectively, for extrapulmonary TB.16 Treatment strategies are individualized and can either involve a splenectomy in cases with enlarged spleens with impending rupture or a more conservative approach with percutaneous drainage and appropriate chemotherapeutic agents (ATT). The success rates of percutaneous drainage for splenic abscess are between 67–100%.17 If surgery is carried out, standard ATT should be taken pre- and postoperatively.
Conclusion
TB should be considered as differential diagnosis in case of isolated splenic abscesses in immunocompetent individuals from endemic
areas. Radiodiagnostic imaging is imperative for faster diagnosis and treatment with percutaneous drainage to defer impending rupture and need for surgical splenectomy.
Disclosure
The authors declared no conflicts of interest. No funding was received for this study.
Acknowledgements
We would like to thank the Department of Radio Diagnosis
and Imaging, PGIMER, Chandigarh.
references
- 1. Alonso Cohen MA, Galera MJ, Ruiz M, Puig la Calle J Jr, Ruis X, Artigas V, et al. Splenic abscess. World J Surg 1990 Jul-Aug;14(4):513-516, discussion 516-517.
- 2. Chun CH, Raff MJ, Contreras L, Varghese R, Waterman N, Daffner R, et al. Splenic abscess. Medicine (Baltimore) 1980 Jan;59(1):50-65.
- 3. Adil A, Chikhaoui N, Ousehal A, Kadiri R. [Splenic tuberculosis. Apropos of 12 cases]. Ann Radiol (Paris) 1995;38(7-8):403-407.
- 4. Singh B, Ramdial PK, Royeppen E, Moodley J, Chetty R. Isolated splenic tuberculosis. Trop Doct 2005 Jan;35(1):48-49.
- 5. Anuradha S, Singh NS, Agarwal SK. Splenic abscess – A diversity within. Indian Academy of Clinical Medicine. 2000;1(3):279-281.
- 6. Paris S, Weiss SM, Ayers WH Jr, Clarke LE. Splenic abscess. Am Surg 1994 May;60(5):358-361.
- 7. Tung CC, Chen FC, Lo CJ. Splenic abscess: an easily overlooked disease? Am Surg 2006 Apr;72(4):322-325.
- 8. Ooi LL, Leong SS. Splenic abscesses from 1987 to 1995. Am J Surg 1997 Jul;174(1):87-93.
- 9. Lee WS, Choi ST, Kim KK. Splenic abscess: a single institution study and review of the literature. Yonsei Med J 2011 Mar;52(2):288-292.
- 10. Sharma SK, Mohan A. Extrapulmonary tuberculosis. Indian J Med Res 2004 Oct;120(4):316-353.
- 11. Ganie FA, Dar GA, Kelam A, Lone H, Dar M, Wani N, et al. Esophageal tuberculosis presenting as dysphagia. Oman Med J 2013;28(3).
- 12. Al-Rikabi AC, Arafah MA. Tuberculosis of the tongue clinically masquerading as a neoplasm: a case report and literature review. Oman Med J 2011 Jul;26(4):267-268.
- 13. Kaur M, Dalal V, Bhatnagar A, Siraj F. Pancreatic tuberculosis with markedly elevated CA 19-9 levels: A diagnostic pitfall. Oman Med J 2016 Nov;31(6):446-449.
- 14. Dixit R, Arya MK, Panjabi M, Gupta A, Paramez AR. Clinical profile of patients having splenic involvement in tuberculosis. Indian J Tuberc 2010 Jan;57(1):25-30.
- 15. Mitra S, Mitra M, Sahoo S, Ghosh BK. Tubercular splenic abscess: Two case reports with review of literature. J Assoc Chest Physicians. 2015;3(2):69-72.
- 16. Hillemann D, Rüsch-Gerdes S, Boehme C, Richter E. Rapid molecular detection of extrapulmonary tuberculosis by the automated GeneXpert MTB/RIF system. J Clin Microbiol 2011 Apr;49(4):1202-1205.
- 17. Thanos L, Dailiana T, Papaioannou G, Nikita A, Koutrouvelis H, Kelekis DA. Percutaneous CT-guided drainage of splenic abscess. AJR Am J Roentgenol 2002 Sep;179(3):629-632.