Kaposi sarcoma (KS) stimulated a renewed interest in vascular tumors in the early eighties due to its association with the acquired immune deficiency syndrome (AIDS). Studies done in Northern Nigeria have shown that as many as 30% of patients with AIDS develop KS, and in many instances diagnosis of the tumor leads to clinical recognition of the syndrome.1 In the absence of human immunodeficiency virus (HIV) infection, KS occurs very rarely in three distinct epidemiologic forms: classic form, endemic form (which is common in some parts of equatorial Africa), and transplant-associated form. Since KS was first described as “idiopathic multiple pigment-sarcoma of the skin” by Moritz Kaposi in 1872, many cases have been reported worldwide.2,3 Before the HIV epidemic, KS showed a greater geographic variation in incidence than almost any other vascular tumor. In the early nineties, it was common in parts of Sub-Saharan Africa such as Uganda and eastern Zaire representing up to 9% of all cancers in men.4 KS was also endemic, although much rarer, in countries around the Mediterranean, particularly Italy, Greece, and the Middle East, but it was almost non-existent elsewhere in the world.5,6 Earlier reports show that KS constitutes 4.2% of all malignant tumors seen in Malawi5,7 and Uganda, and in mainland Tanzania it accounts for about 4% of all histologically verified malignancies.4,5,7 Furthermore, in a previous report in Zaria, Nigeria, it accounted for 1.3% of all malignancies and 77.8% of all malignant vascular lesions.8 In a study of 148 soft tissue sarcomas in Ilorin, Nigeria, 8.1% were KS.9
We sought to establish the frequency and histological patterns of KS in Benin City and its environs with the intention of reminding clinicians how common these lesions are particularly in this era of HIV/AIDS. Data derived from this research would serve as baseline data for management of patients and further study.
Methods
We conducted a retrospective analysis of all histologically confirmed benign, intermediate, and malignant vascular neoplasms in the Department of Pathology, University of Benin Teaching Hospital, Benin City, between January 1994 and December 2013. The pathology request cards were retrieved to obtain data on age, sex, clinical features, type of biopsy, and diagnosis. We also recorded a description of the gross morphology and location of individual neoplasms.
The routine hematoxylin and eosin (H/E) stained slides of specimens were retrieved from the Pathology Department records and histologically examined. Where the glass slides were missing or broken, fresh sections were taken and stained from stored original tissue blocks. Special histochemical stains such as Masson’s trichrome were used to outline the basic framework of the tumor while reticulin stain was used to outline collagen bundles, perivascular tumors, and to differentiate them from aggregates of epithelial tumors.
The age and sex frequency distribution of KS were analyzed. Tumor types and patterns distribution were also analyzed. The vascular tumors were classified into benign, intermediate, and malignant categories according to the World Health Organization (WHO) 2013 criteria with emphasis on KS as an intermediate vascular tumor. The histological types and patterns of KS were also examined and analyzed by two experienced consultant dermato-pathologists. The absence of immunohistochemistry of KS-associated herpes virus-8 nuclear antibodies for confirmation of H/E diagnosis made way for the exclusion of few controversial cases that would have been included in this study.
Data were analyzed using simple statistical procedures and results were converted to relative values in frequency tables.
Ethical approval was duly obtained from the ethical committee of the University of Benin Teaching Hospital, Benin City before the commencement of this work.
Results
In this 20-year study period, 269 histologically diagnosed vascular tumors were scrutinized and analyzed. Of these cases, benign tumors constituted 180 cases accounting for (66.9%) while borderline and malignant tumors constituted 30.9% (n = 83) and 2.2% (n = 6), respectively. KS thus constituted 79 (95.2%) out of the 83 borderline vascular tumors. Out of the 79 cases of KS, 76 (96.2%) cases were confirmed HIV/AIDS patients already on highly active antiretroviral therapy.
Table 1: Age and ex distribution of Kaposi sarcoma.
|
1–9 |
1/2 |
3 (3.8) |
|
10–19 |
1/1 |
2 (2.5) |
|
20–29 |
3/6 |
9 (11.4) |
|
30–39 |
19/14 |
33 (41.8) |
|
40–49 |
8/6 |
14 (17.7) |
|
50–59 |
8/5 |
13 (16.5) |
|
60–69 |
1/2 |
3 (3.8) |
|
70+ |
0/2 |
2 (2.5) |
Table 1 shows the overall age and sex distribution of KS. It had a peak incidence in the fourth decades with almost equal distribution of incidence on either side of this peak. The age range for KS was from six months to 79 years; however, two cases were seen in cervical lymph node in the pediatric age brackets of six months and four years, respectively. KS accounted for 41 males and 38 females, respectively, giving a male to female ratio of 1.1:1.0 The mean age for KS over this period of study was 39.2±3.6 years. There was a significant variation in the yearly trend of KS [Figure 1]. This yearly trend remained relatively stable from 1994 to 2004. Thereafter, there was a sudden sharp rise in trend from 2005 with a peak in 2008. This was followed by a slight fairly stable decline from 2008–2013.
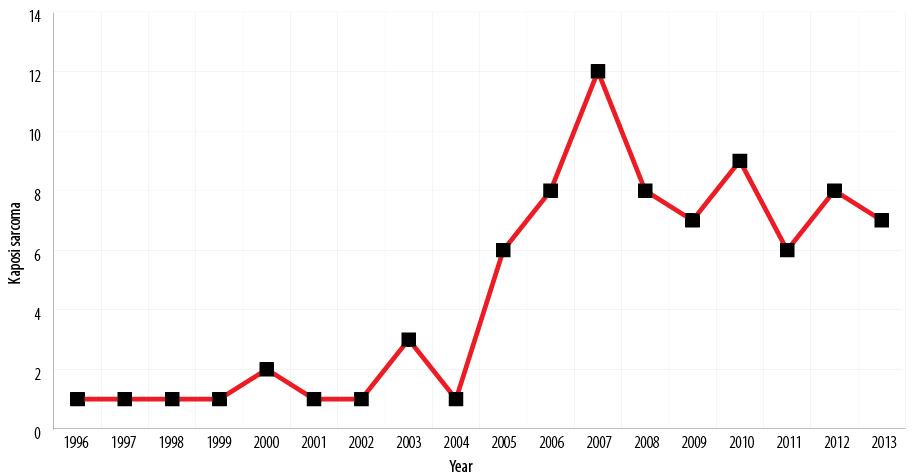
Figure 1: Yearly trend of Kaposi sarcoma cases from 1994 to 2013.
The leg and foot region was the most common site of occurrence of KS [Table 2]. In all 79 cases, 38 (48.1%) occurred in the leg and foot. The second majority site of occurrence of KS was in the trunk accounting for 25 cases (31.6%). Eight cases of KS (10.1%) occurred in the forearm and hand while three cases (3.8%) occurred in the anogenital region. KS in the head and neck accounted for three cases (3.8%) while only two cases (2.5%) were encountered in the cervical lymph node.
Table 3 shows the pathological stages and phases of KS. The most common stage of KS observed in this study was the nodular stage (48.1%). Furthermore, the most common phase of KS was mixed phase constituting 50.6%.
Table 2: Anatomic site distribution of Kaposi sarcoma.
|
Head and neck |
3 (3.8) |
|
Leg and feet |
38 (48.1) |
|
Forearm and hand |
8 (10.1) |
|
Trunk |
25 (31.6) |
|
Anogenital |
3 (3.8) |
|
Cervical lymph node |
2 (2.5) |
Discussion
The most common borderline vascular tumor was KS. Accordingly, KS accounted for 95.2% of borderline vascular tumors. This is in tandem with a previous similar study where KS constituted 91% of all borderline and malignant vascular tumors.10 However, a series of reports have documented that KS showed a greater geographic variation in incidence than almost any other borderline tumor. Earlier reports have documented that it was commonly seen in parts of Sub-Saharan Africa, such as Uganda and Zaire, constituting 9% of all cancer in men. In all of these areas, KS was considerably more common in men than women.4,5,7,11 Nevertheless, parts of Africa with a high prevalence of HIV/AIDS and where KS was relatively common even before the era of AIDS have seen an explosion in the incidence of the disease in the last decade. Previous studies have reported that in the past 10–15 years, the incidence of KS has increased about 20-fold in Uganda and Zimbabwe such that it is now the most common borderline and malignant tumor in men and the second most common in women.4,5,7,11 Similarly, between 1988 and 1996, the incidence of KS has risen at least three-fold in South Africa and continued to increase as the HIV epidemic grew.11
Table 3: Histopathological stages and phases of Kaposi sarcoma.
|
Stage |
|
|
Patch |
27 (34.2) |
|
Plaque |
14 (17.7) |
|
Nodular |
38 (48.1) |
|
Total |
79 (100) |
|
Phases |
|
|
Anaplastic |
16 (20.3) |
|
Monomorphic |
23 (29.1) |
|
Mixed |
40 (50.6) |
A male to female ratio of 1.1:1.0 for KS was seen in this study. This is slightly similar to earlier reports from this same center where the male to female ratio was 1.7:1.12 However, our report is more recent and showed a close gap in the male to female ratio. This once more correlates with more recent epidemiological data showing a closed gap in the trend of previously overwhelming preference for the male gender by this tumor in the pre-AIDS era. For example, data from the South African National Cancer Registry shows that between 1992 and 1996, the incidence rates of KS doubled in men but increased about seven-fold in women, such that the sex ratio of 7:1 in males versus females in 1988 has now declined to only 2:1.13
In this 20-year study, two cases of KS were reported in the pediatric age group and both were seen in the cervical lymph nodes. This finding, though rare, is not new finding lymphadenopathic (endemic) KS also rarely affect the pediatric age groups and run a more aggressive course.14
In this study, KS constituted the most common borderline vascular tumor with a sharp rise in the last decade. This accounted for a six-fold rise in the incidence of KS from 2005 to 2008. This is supported by reports of other researchers where a three- to 20-fold rise in the incidence of KS was documented.4,11,13,15 Again within the past decade, a 20-fold rise in the prevalence of KS was documented in Zimbabwe and Uganda. This thus supports the fact that it remained the most common malignancy in both countries at that time.11,13,15 Studies have documented that KS has the lowest incidence in Asia and Caucasians while the highest incidence is seen in blacks.2,15 The reason for the high incidence in blacks is partly due to high increase of KS-associated herpesvirus (KSHV-8) in Africa compared to its low incidence in Asia sub-continent.16,17 This high incidence of KS in these sub-Saharan African countries is due to the high rate of KSHV-8 infection in their general population.16–18 The mean age for KS was 39.2 years in this study. This report is slightly variable from a report by Pitche et al,18 where the mean age was 35.2 years. Nevertheless, this may partly be supported by the fact that both peak ages correspond to peak ages of sexual activities in both males and females as the majority of AIDS patients globally contract HIV through sexual means.
The most common anatomic site of occurrence was in the leg and feet. Globally, similar studies have also corroborated this finding.15–18 No reason was attributed to this finding. In our study, the leg and foot were the most common site accounting for 48.1% of KS cases. This finding is not far from reports in Jos, Northern Nigeria, where the leg and foot were the most common site constituting 58.5% of all anatomic site distribution of KS.19 This report is also similar to reports of other Nigerian and African researchers. Nevertheless, the reason for this predilection for the leg and foot cannot be readily ascertained.
In this study, the most prevalent histological type was the mixed KS which has both histopathological features of both anaplastic and monomorphic KS. This thus constituted 50.6% of all histological types. This is similar to the report from Northern Nigeria where the mixed KS was the most common type and constituted 61.5% of all histological types.19 Furthermore, the most commonly observed pattern in the study was the nodular pattern, which constituted 48.1%. This again is similar to other researchers reports globally.6,19 The reason for this high prevalence of nodular pattern is partly because most patients present late to the hospital. This may be due to poverty, ignorance, and stigmatization associated with AIDS. Some of these patients may prefer alternative therapy to medical management.
Conclusion
KS was the most common borderline vascular tumor, and the majority of cases were associated with HIV/AIDS. The mean age of presentation was seen in the fourth decade of life. There was a sharp rise in its incidence in this last decade. The mixed KS and nodular KS were the most frequently encountered type and pattern of KS encountered, respectively.
Disclosure
The authors declared no conflicts of interest. No funding was received for this study.
references
- 1. Kagu MB, Nggada HA, Garandawa HI, Askira BH, Durosinmi MA. AIDS-associated Kaposi’s sarcoma in Northeastern Nigeria. Singapore Med J 2006 Dec;47(12):1069-1074.
- 2. Bradford PT. Skin cancer in skin of color. Dermatol Nurs 2009 Jul-Aug;21(4):170-178.
- 3. Adeyi O, Banjo AA. Malignant Tumors of the Skin. A six year review of histological diagnosed cases (1990-1995). Nig Q J Hosp Med 1996;2:99-102.
- 4. Wabinga HR, Parkin DM, Wabwire-Mangen F, Mugerwa JW. Cancer in Kampala, Uganda, in 1989-91: changes in incidence in the era of AIDS. Int J Cancer 1993;54(1):26-36.
- 5. Greenlee RT, Murray T, Bolden S, Wingo PA. Cancer statistics, 2000. CA Cancer J Clin 2000 Jan-Feb;50(1):7-33.
- 6. Rosai J. Skin tumours and tumour-like conditions. In: Rosai J, editor. Rosai and Ackerman’s surgical pathology. 9th ed, Mosby Philadelphia; 2004. p. 130-245.
- 7. Bassett MT, Chokunonga E, Mauchaza B, Levy L, Ferlay J, Parkin DM. Cancer in the African population of Harare, Zimbabwe, 1990-1992. Int J Cancer 1995 Sep;63(1):29-36.
- 8. Rafindadi AH. Childhood vascular tumours in Zaria, Nigeria. West Afr J Med 2000 Apr-Jun;19(2):101-104.
- 9. Adeniji KA. Histopathological and histochemical patterns of soft tissue sarcoma Nig. Qt J Hosp Med 2000;10(3):192-197.
- 10. Obaseki DE, Ahiwu WO, Aligbe JU, Igbe AP, Eze GI, Forae GD. Morphological patterns of vascular tumours in Benin-City, Nigeria: A twelve year retrospective review. Niger J Surg Sci 2013;23(1):9-13.
- 11. Sitas F, Newton R. Kaposi’s sarcoma in South Africa. J Natl Cancer Inst Monogr 2001;28(28):1-4.
- 12. Onunu AN, Okoduwa C, Eze EU, Adeyekun AA, Kubeyinje EP, Schwartz RA. Kaposi’s sarcoma in Nigeria. Int J Dermatol 2007 Mar;46(3):264-267.
- 13. Sitas F, Madhoo J, Wessie J. Incidence of histologically diagnosed cancer in South Africa, 1993–1995. National Cancer Registry of South Africa. Johannesburg: South African Institute of Medical Research; 1998.
- 14. Slavin G, Cameron HM, Forbes C, Mitchell RM. Kaposi’s sarcoma in East African children: a report of 51 cases. J Pathol 1970 Mar;100(3):187-199.
- 15. Forae GD, Olu-Eddo AN. Malignant skin tumors in Benin-City, South-South, Nigeria. Oman Med J 2013 Sep;28(5):311-315.
- 16. Alzahrani A, El-Harith EA, Milzer M. Increased seroprevalence of human herpes virus-8 in renal transplant recipients in Saudi Arabia. Nephrology Dialysis Transplant, 2005;20(11):2532-2536.
- 17. Antman K, Chang Y. Kaposi’s sarcoma. N Engl J Med 2000 Apr;342(14):1027-1038.
- 18. Pitche P, Kombate K, Napo-Koura G, Mijiyawa M, Tchangai-Walla K. [Kaposi sarcoma in dermatology consultations in Lome. Togo]. Med Trop 1995;55(3):246-248.
- 19. Mandong BM, Chirdan LB, Anyebe AO, Manasseh AN. Histopathological study of Kaposi sarcoma in Jos: A 16 year review. Ann Afr Med 2004;3:174-176.