|
Abstract
Prevalence of persistent endometriosis in women after menopause without any hormonal replacement therapy is very rare. This is a case of a woman with previous history of total hysterectomy and bilateral salpingo-oophorectomy for endometriosis who presented with hemoperitoneum, vaginal bleeding, pelvic mass, and pulmonary thromboembolism mimicking as rectovaginal septum carcinoma. This is the first case report with a unique mode of presentation wherein the patient presented with hemoperitoneum requiring emergency embolization of the vessel to stabilize the patient. She underwent en bloc resection of the tumor with high anterior resection of the rectum. Histopathology confirmed endometriosis.
Keywords: Endometriosis; Ovarian cancer; Menopause.
Introduction
The incidence of endometriosis in post-menopausal women is 2% to 5%1,2 and is commonly seen in those who have received hormonal replacement therapy after menopause.3 This report is about a rare case of a patient with post-menopausal endometriosis, who presented with intra-abdominal bleeding, pelvic mass, vaginal bleeding and pulmonary thromboembolism. With clinical and radiological findings and past history of endometriosis, the disease was considered as rectovaginal septum carcinoma of clear cell type resulting from malignant transformation of endometriosis.
Case Report
A 50-year-old para 2 was referred to our hospital in view of pelvic mass, bilateral hydronephrosis and pulmonary embolism. She had initially presented to a private hospital with a history of acute abdomen due to hemoperitoneum and bleeding per-vagina. A contrast enhanced CT scan of the abdomen and pelvis showed large hemoperitoneum with solid dense areas in the caudal portion of the pelvis with a blush of increasing contrast enhancement in the central part of the pelvis indicating active bleeding likely from a branch of inferior mesenteric artery (Figs. 1A and B). She underwent emergency embolization of the feeding vessels with gelfoam to stop the active bleeding. Following the embolization, CT showed a lobulated soft tissue density mass in the pelvis measuring about 15 × 6.6 × 6.5 cm which was compressing the rectum and the bladder and with bilateral hydronephrosis (Figs. 2A and B). Serum CA-125 was 595.8 and CA-19.9 was 26.9.
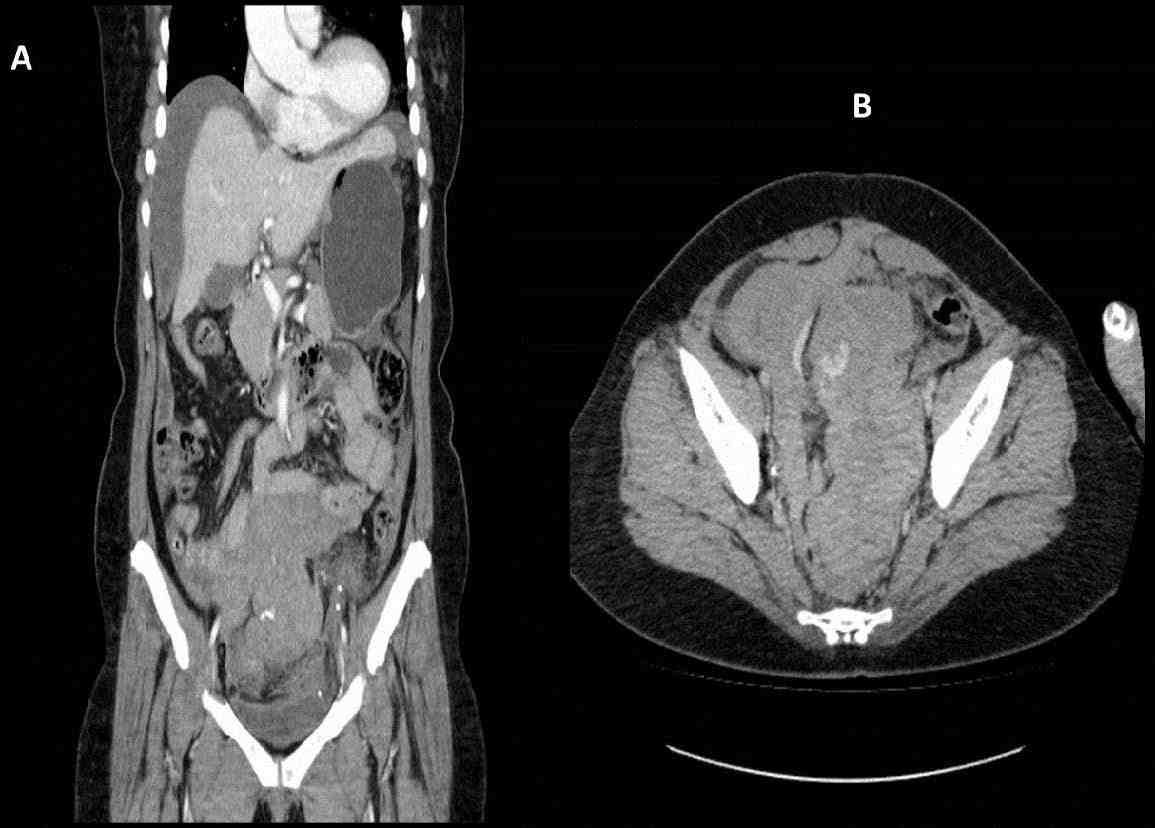
Figures 1A and B: Computed tomography (CT) of abdomen, pelvic and chest. A: Large amount of dense free fluid within the pelvis and moderate amount of more hypodense free fluid is also visualized in the abdomen. B: There is a blush of increasing contrast enhancement in the central and right hemipelvis in keeping with active hemorrhage.
Her past surgical history suggested that she had undergone total hysterectomy for fibroid uterus and after two years she had bilateral salpingo-oophorectomy and partial vaginectomy for deeply infiltrating endometriosis. Post-operatively, she was neither treated for her endometriosis nor was she on any hormonal replacement therapy. With this mode of clinical presentation, the differential diagnosis considered were carcinoma of the peritoneum or recto-vaginal septum due to malignant transformation of the remnant endometriosis mainly of clear cell type due to the typical features of pelvic mass, pulmonary embolism and past history of endometriosis.
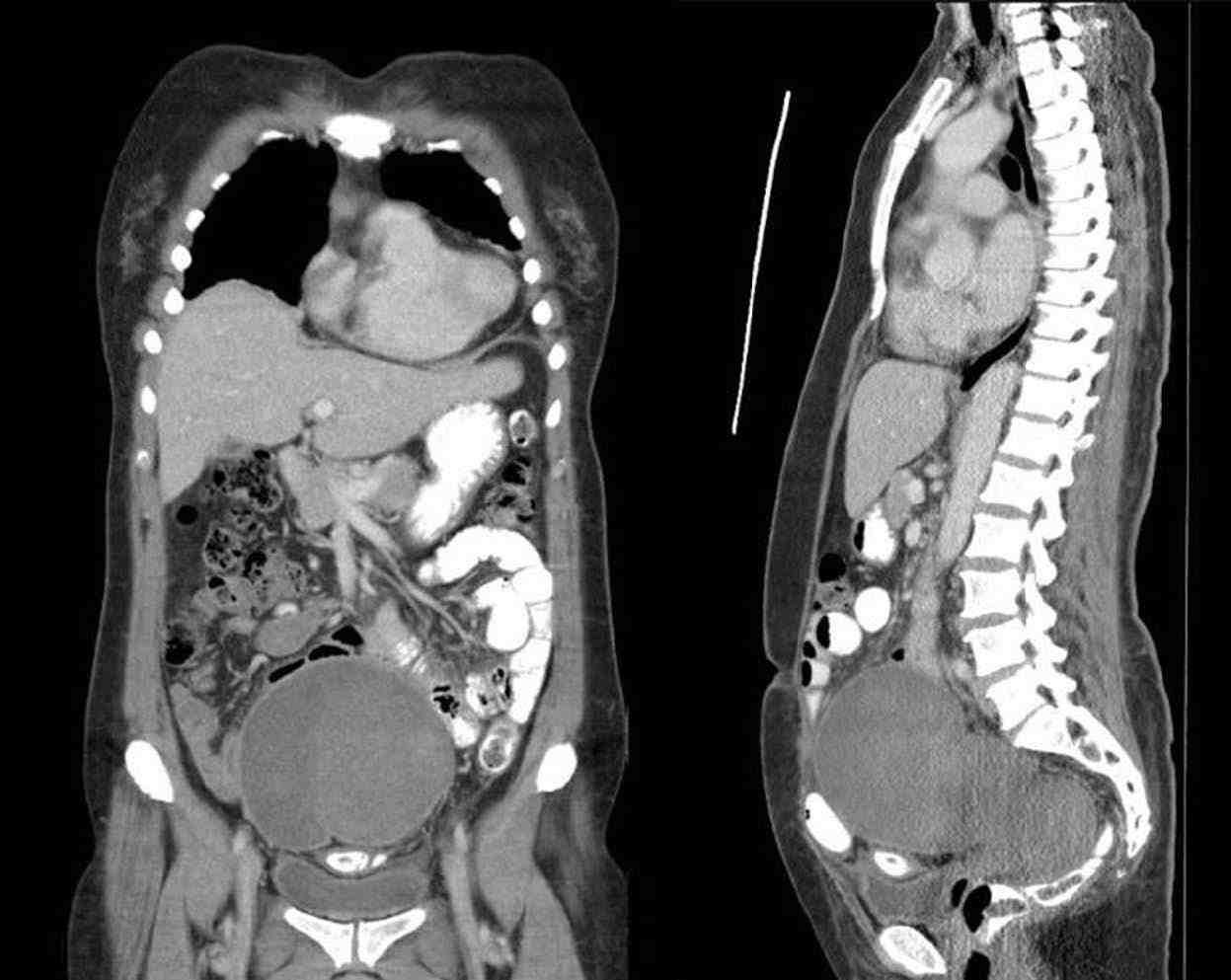
Figures 2A and B: CT of abdomen, pelvis and chest after embolization of inferior mesenteric artery: Lobulated soft tissue density mass seen in the pelvis measuring about 15 × 6.6 × 6.5 cm, compressing the rectum and bladder and displacing the small bowel loops laterally.
She underwent laparotomy revealing a 5 × 3 × 2.5 cm mass which was densely adherent to the rectosigmoid colon, to the pelvic side walls involving bilateral ureters, and eroding into the vagina causing ulceration of the vaginal vault. There was neither free intraperitoneal fluid nor any signs of pelvic or abdominal endometriosis. Preoperative CT showed a 15 × 6.6 × 6.5 cm mass in the pelvis, but intraoperatively, only a 5 × 3 × 2.5 cm mass was noted in the pelvis giving rise to a possibility that the intraperitoneal bleeding had undergone resorption. She underwent uretrolysis, en bloc resection of the tumor, and high anterior resection of the rectum. Postoperatively, the patient made uneventful recovery and was discharged home on the 9th postoperative day. Final histopathological findings showed features consistent with so-called necrotic pseudoxanthomatous nodule, associated with longstanding endometriosis and no features of malignancy.
Discussion
It has been hypothesized that endometriosis is estrogen-dependent and that progesterone inhibits the cellular proliferation, therefore endometriosis is commonly seen in women of reproductive age group and very rarely seen after menopause. Different theories have been postulated for recurrence of endometriosis in women who have undergone surgical menopause, such as ovarian remnant syndrome (ORS), wherein part of the ovarian tissue has been left behind after bilateral oophorectomy which continues to produce hormone and stimulate the ectopic endometrial implant. The endometrial implants can also be reactivated by exogenous estrogen in the form of hormonal replacement therapy,3,4 or endogenous estrogen which comes from peripheral conversion of androgen and androstenedione from adrenal glands. Evidence also shows that endometrial implants harbor aromatase expression which is stimulated by PGE2 leading to local production of estrodiol. These implants also lack hydroxysteroid dehydrogenase (17β-HSD) type 2 expression thus impairing conversion of estradiol to estrone which results in local accumulation of potent estrodiol. Another possible hypothesis of endometriosis after menopause is due to spontaneous coelomic metaplasia and vascular endometrial cell transportation. It is possible that our patient had ovarian remnant syndrome because her BMI was normal, had no history of HRT but had past history of deep endometriosis making it possible that part of ovarian tissue must have been left in-situ during laparotomy done for bilateral salpingo-oophorectomy and vaginectomy. Since we did not have preoperative serum estradiol and FSH levels, it was difficult to demonstrate her true menopausal status.
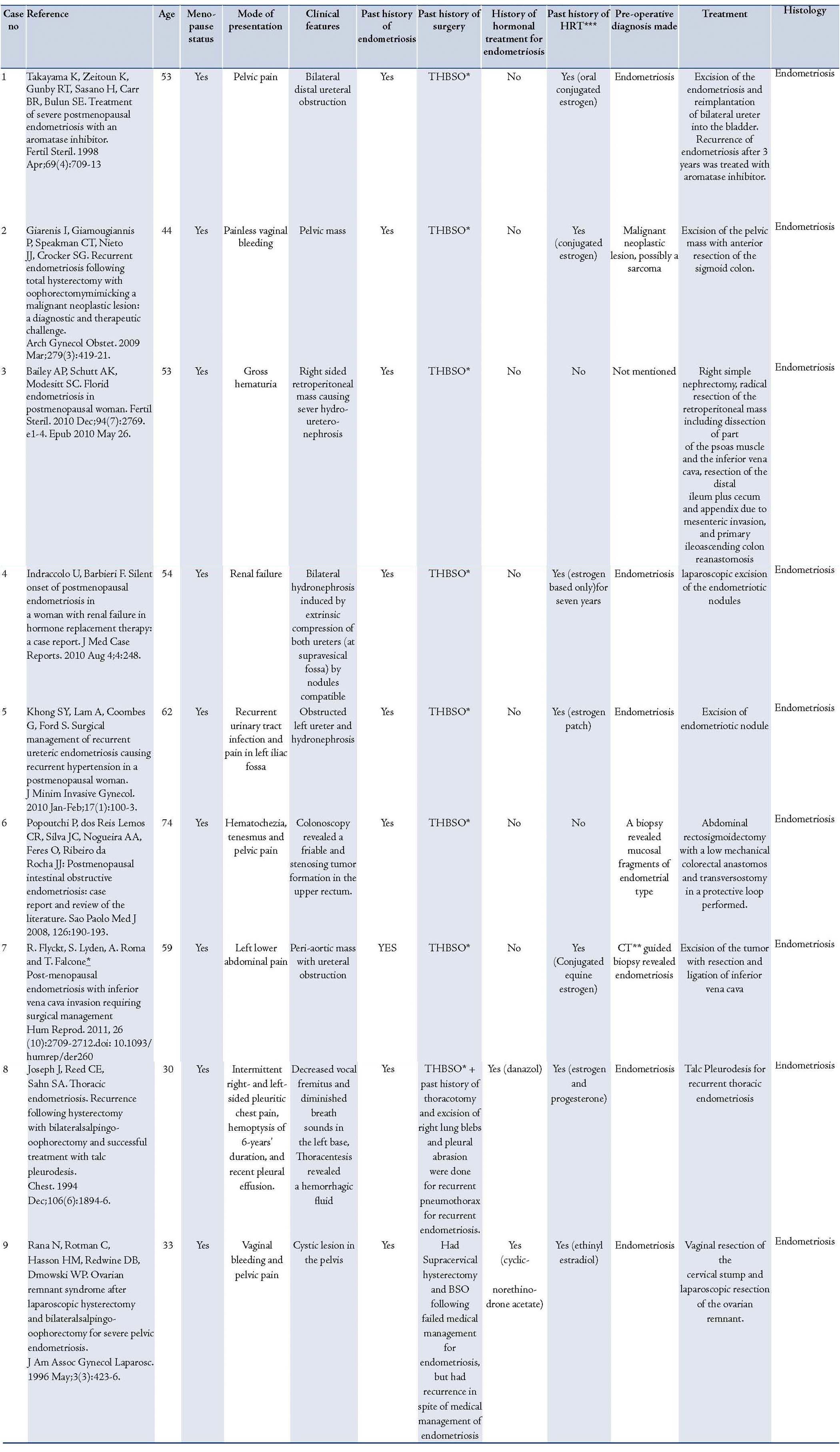
Table 1 shows a summary of studies on endometriosis in women who have undergone surgical menopause. Presentation of endometriosis in post-menopausal patients can be unpredictable and mainly depends on the location of the endometriotic implants. Since endometriosis is usually associated with surrounding inflammation and fibrosis, symptoms can sometimes be very severe. Since the time of Sampson in 1925,5 numerous case reports and review of the literature have shown that endometriosis can be a precursor of ovarian, primary peritoneal or recto-vaginal septum cancer mainly of clear cell, endometroid type.6,7 Literature has also shown that endometriosis can act as a precursor for both cystic and adenofibromatous types of clear cell carcinoma of the ovary.8 Endometriosis can sometimes present with ascites, pelvic mass and pleural effusion mimicking as advanced ovarian cancer,9 thus making preoperative diagnosis of endometriosis, especially in women who have undergone total hysterectomy and bilateral salpingo-oophorectomy, very challenging to the treating physician. Clear cell carcinoma demonstrates unique clinical features such as large pelvic mass, thromboembolic complications and hypercalcemia.
In this case report, the patient had clinical features of pelvic mass, hemoperitoneum and pulmonary embolism and our diagnosis was carcinoma of the recto-vaginal septum of clear cell type associated with endometriosis. Surgical resection of all endometriotic implants and restoring normal anatomy is the treatment of choice. Hence, in patients with deeply infiltrating endometriosis, radical surgery involving bowel resection becomes inevitable. Following surgery postoperative hormonal treatment to suppress endometriosis has not yet been established.10
Table 1: Summary of studies of endometriosis in women after total hysterectomy with bilateral salpingo-oophorectomy (THBSO).
Conclusion
Endometriosis after surgical menopause is rare and symptoms can vary based on the site of endometriotic implants. Sometimes endometriosis can present with symptoms which can mimic pelvic malignancies and also since endometriosis confers the risk of malignant transformation, this makes it difficult for a treating physician to make appropriate pre-operative diagnosis. Although endometriosis after surgical menopausal is rare, it should be considered in the differential diagnosis of abdominal or pelvic mass, especially in women with past history of endometriosis.
Acknowledgements
We would like to thank Professor Soo Khee Chee and A / Prof Koong Heng Nung for their help with the design of the study protocol. No conflict of interests or funding to declare.
References
1. Manero MG, Royo P, Olartecoechea B, Alcázar JL. Endometriosis in a postmenopausal woman without previous hormonal therapy: a case report. J Med Case Rep 2009;3:135.
2. Punnonen R, Klemi PJ, Nikkanen V. Postmenopausal endometriosis. Eur J Obstet Gynecol Reprod Biol 1980 Dec;11(3):195-200.
3. Matorras R, Elorriaga MA, Pijoan JI, Ramón O, Rodríguez-Escudero FJ. Recurrence of endometriosis in women with bilateral adnexectomy (with or without total hysterectomy) who received hormone replacement therapy. Fertil Steril 2002 Feb;77(2):303-308.
4. Fedele L, Bianchi S, Raffaelli R, Zanconato G. Comparison of transdermal estradiol and tibolone for the treatment of oophorectomized women with deep residual endometriosis. Maturitas 1999 Aug;32(3):189-193.
5. Munksgaard PS, Blaakaer J. The association between endometriosis and ovarian cancer: a review of histological, genetic and molecular alterations. Gynecol Oncol 2012 Jan;124(1):164-169. Published online 26 Oct 2011.
6. Munksgaard PS, Blaakaer J. The association between endometriosis and ovarian cancer: a review of histological, genetic and molecular alterations. Gynecol Oncol 2012 Jan;124(1):164-169. Published online 26 Oct 2011.
7. Mabrouk M, Vicenzi C, Ferrini G, Geraci E, Forno SD, Caprara G, et al. Mixed adenocarcinoma of the rectovaginal septum associated with endometriosis and endometrial carcinoma: a case report. Case Rep Oncol 2011 Jan;4(1):149-154. Published online 18 Mar 2011.
8. Cakir E, Aydin E, Durmus NI, Samdanci E, Sahin N, Nurkabul Z. Prımary Ovarıan Clear Cell Adenofıbroma of Borderlıne Malıgnancy. Oman Med J 2012;27(1) online publication.
9. Goumenou A, Matalliotakis I, Mahutte N, Koumantakis E. Endometriosis mimicking advanced ovarian cancer. Fertil Steril. 2006 Jul;86(1):219.e23-5.
10. Yap C, Furness S, Farquhar C. Pre and post operative medical therapy for endometriosis surgery. Cochrane Database Syst Rev 2004;(3):CD003678. doi:10.1002/14651858.CD003678.pub2.
|