Beta-thalassemia syndromes are a heterogeneous group of autosomal recessive hereditary anemias resulting from defective synthesis of β-globin chains of adult hemoglobin (Hb) A. This defect can lead to a wide spectrum of clinical phenotypes ranging from severe anemia to asymptomatic presentations.1,2 The Thalassemia International Federation guidelines (2022)3 have reclassified thalassemia into two major groups based on clinical severity and transfusion requirements: transfusion-dependent thalassemia (TDT) and non-transfusion dependent thalassemia (NTDT).3
Hypercoagulability has been identified in thalassemia, particularly in NTDT. This hypercoagulability is multifactorial, involving abnormal platelets and pathological red blood cells (RBCs) as key contributors to thrombotic events. Oxidative stress of thalassemic RBCs is associated with the expression of negatively charged phospholipids on their outer membrane leaflet, which can increase thrombin generation.4,5
In response to stress, apoptosis, or activation, cells release extracellular vesicles (EVs). These are submicrometric bioactive membrane-bound particles, including apoptotic bodies, microparticles (MPs), and exosomes, which can be classified by size. MPs are larger EVs that bud off from the plasma membrane; 70–90% being platelet-derived.6 In hematological disorders such as thalassemia, they may have the potential to be prognostic biomarkers for hypercoagulability.7
Despite having acceptable Hb levels and being transfusion-independent, patients with NTDT are more prone to thalassemia-related complications than those with TDT. Thromboembolic events—such as deep vein thrombosis, portal vein thrombosis, pulmonary embolism, and cerebral thrombosis—are more common in NTDT and typically manifest at older ages.8 The prevalence of thrombotic events can reach up to 20% in NTDT patients, compared with < 1% in TDT patients.9 The risk of thrombosis, including silent thrombosis, is significantly increased by splenectomy in thalassemia patients, due to decreased clearance of damaged RBCs, chronic platelet activation, and increased MP shedding.10,11
Despite the increasingly recognized role of echocardiography as an initial screening tool to suspect pulmonary hypertension (PH), cardiac catheterization remains the gold standard.12 Additional markers include the amino-terminal fragment of proB-type natriuretic peptide (NT-proBNP),13 a hormone released in response to cardiomyocyte stretching. In the early phases of cardiac involvement, NT-proBNP level increases before an increase in diastolic pressure. Elevated NT-proBNP is a strong indicator of the presence of PHT in thalassemia patients.14
The present study aims to evaluate the levels of circulating red cell- and PMPs in a cohort of Egyptian children with NTDT and their possible contribution to the disease-associated hypercoagulability.
Methods
This case-control study included 50 children with NTDT, attending the Pediatric Hematology outpatient clinic of a tertiary teaching Hospital in Alexandria, Egypt, from November 2022 to May 2023, along with 50 sex- and age-matched healthy controls. Inclusion criteria for the NTDT group were: age 8–16 years, clinical presentation after two years old, mild to moderate anemia, mild hepatomegaly or splenomegaly; fewer than six RBC transfusions in the preceding six months, diagnostic confirmation via blood counts, Hb electrophoresis, and (when available) PCR β-globin gene analysis.15 We excluded children who received blood transfusion in the past three months, had hepatitis C positivity, or had a family history of thrombotic or bleeding disorders. All data were extracted from the hospital medical records.
Ethical approval for the study was given by the Ethics Committee of the Faculty of Medicine, Alexandria University, Egypt, IRB No. 0001298, FWA No. 00018699; EC No. 0201580 on November 18, 2021. Written informed consent was obtained from the parents or legal guardians of all participating children.
All participants underwent a detailed medical history taking and clinical examination, especially for chest and cardiac signs, hepatic size and splenic status, and lower limb signs of thrombosis. Routine laboratory investigations included complete blood count, serum chemistry, serum ferritin level (measured using Centaur XPT Siemens automated analyzer), and D-dimer (Sysmex Siemens automated analyzer). A D-dimer level of ≥ 550 ug/L was considered to indicate a hypercoagulable state.16 Platelet aggregation was assessed using an aggregometer with adenine 5-diphosphate as agonist at 1:10 dilution. Using monoclonal antibodies (mAbs), NT-proBNP levels were quantitatively determined by enzyme-linked immunosorbent assay.
Microparticle Analysis.17 Venous blood (2 mL) was collected from each patient using atraumatic technique into a 3.2% sodium citrate tube and processed within 15 minutes of collection. MPs were isolated by centrifuging whole blood at 1500 × gravity (g) for 15 min at room temperature to obtain platelet-poor plasma. The residual plasma above the cell pellet was discarded, and platelet-poor plasma further centrifuged at 14 000 × g for 2 minutes at 20 °C to yield platelet-free plasma. This was centrifuged again at 14 000 × g for another 45 minutes at 4 °C. The supernatant was discarded, and the MP pellet was resuspended in phosphate-buffered saline and vortexed.
A 30 µL aliquot of platelet-free plasma was incubated with appropriate amounts of specific mAbs and 10 µL of Annexin V. The mAbs included anti-glycophorin A (GPA; CD235a-FITC) for identification of RBC–derived MPs and anti-glycoprotein IIb/IIIa (CD41a-PE) for identification of PMPs. All mAbs were purchased from BD Biosciences (San Jose, CA, USA. Megamix-Plus FSC beads (BioCytex) beads were used to define MPs in the 0.3–1.0 μm size range. Platelet- (PMPs) and erythrocyte- (Ery-MPs), were defined as AnnV+/CD41+, AnnV+/CD235a+, respectively). After Ab labeling, the samples were acquired on BD FACSCanto II flow cytometer and the resultant data were analyzed using FSCSDiva software (BD biosciences). Flow cytometric plots of MPs of a representative sample is presented in Figure 1.
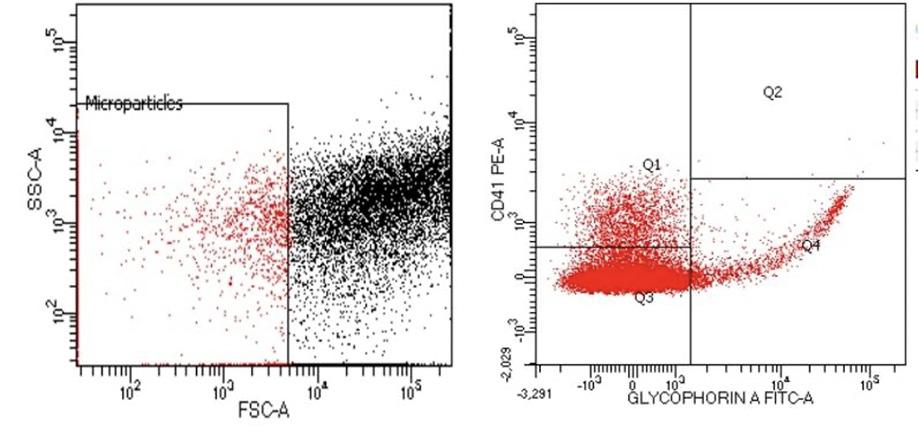 Figure 1: Flowcytometry analysis of erythroid derived microparticles (CD235a+) and platelet derived microparticles (CD 41+).
Figure 1: Flowcytometry analysis of erythroid derived microparticles (CD235a+) and platelet derived microparticles (CD 41+).
Statistical analysis was carried out using IBM SPSS Statistics for Windows, version 25 (IBM Corp., Armonk, N.Y., USA). The Shapiro–Wilk test was used to assess the normality of distribution. Normally distributed variables were expressed as mean ± SD and compared using the independent t-test, while the non-normally distributed data were presented as medians and compared using the Mann-Whitney U-test.18
A prediction nomogram for hypercoagulability was constructed using the Orange Data Mining Toolbox software,19 based on three key independent predictors, PMPs, EMPs, and ferritin. Sample size calculations targeted a power of study of 80% with β error of 20% and an alpha level of 5% (significance: 95%). Statistical significance was set at p < 0.05.20
Results
The study included 50 NTDT patients (32 boys and 18 girls) aged 8–16 years (mean: 10.4 ± 2.9). The control group comprised 50 apparently healthy children, matched for age (p = 0.701) and sex (p = 0.309). None of the NTDT patients had clinical evidence of PH or thrombosis, but six patients (12.0%) underwent splenectomy.
The demographic and laboratory parameters of the two groups are presented in Table 1. NTDT patients showed significantly lower Hb levels, RBC counts, and significantly higher reticulocyte counts compared to controls (p < 0.001). There was no significant difference in platelets count (p = 0.325); however, nine patients (18%) had thrombocytosis (platelets > 450 × 106/µL). Levels of EMP, PMP, and serum ferritin were significantly higher in NTDT group (p < 0.001). NT-proBNP was also higher in NTDT patients, but the difference did not reach statistical significance (p = 0.081).
Table 1: Comparison of children with non-transfusion dependent thalassemia (NTDT) (N = 50) and healthy control group (N = 50).
|
Age, years
|
|
Min-Max
|
8–16
|
8–16
|
t(W)(df=87.850) = 0.385
p = 0.701
|
|
Mean ± SD
|
10.4 ± 2.9
|
10.6 ± 2.0
|
|
Sex, %
|
|
Male
|
32 (64.0)
|
27 (54.0)
|
χ2(df=1) = 1.033
p = 0.309
|
|
Female
|
18 (36.0)
|
23 (46.0)
|
|
Hemoglobin, g/dL
|
|
Min-Max
|
5.2–11.9
|
10.30–12.6
|
t(W)(df=70.315) = 20.635
p < 0.001*
|
|
Mean ± SD
|
7.5 ± 1.2
|
11.42 ± 0.6
|
|
Red blood cell count, ×106/μL
|
|
Min-Max
|
2.4–5.7
|
4.5–5.4
|
t(W)(df=58.661) = 11.208
p < 0.001*
|
|
Mean ± SD
|
3.8 ± 0.7
|
4.9 ± 0.2
|
|
Reticulocytes, %
|
|
Min-Max
|
0.5–6.8
|
0.1–2.0
|
t(W)(df=67.2001) = 13.723
p < 0.001*
|
|
Mean ± SD
|
3.7 ± 1.4
|
0.7 ± 0.6
|
|
Platelets count, ×106/μL
|
|
Min-Max
|
146.0–1153.0
|
190.0–437.0
|
t(W)(df=62.585) = 0.992
p = 0.325
|
|
Mean ± SD
|
330.2 ± 207.7
|
299.1 ± 78.1
|
|
Serum Ferritin, µg/L
|
|
Min-Max
|
17.9–2086.0
|
20.00–62.8
|
t(W)(df=49.076) = 6.621
p < 0.001*
|
|
Mean ± SD
|
441.4 ± 433.9
|
35.0 ± 12.1
|
|
Erythrocyte-derived microparticles, %
|
|
Min-Max
|
0.3–20.0
|
0.8–3.3
|
t(W)(df=52.428) = 3.898,
p < 0.001*
|
|
Mean ± SD
|
4.5 ± 4.7
|
1.9 ± 0.8
|
|
Platelets-derived microparticles, %
|
|
Min-Max
|
1.0–28.1
|
0.9–3.3
|
t(W)(df=51.652) = 6.281
p < 0.001*
|
|
Mean ± SD
|
6.1 ± 4.4
|
2.1 ± 0.7
|
|
N-Terminal Pro-B-type natriuretic peptide, pg/mL
|
|
Min-Max
|
64.5–142.0
|
54.46–122.6
|
W: welch’s t-test; χ2: pearson chi-square; df: degree of freedom; * Significant.
Table 2 shows a significant positive correlation between PMPs and platelet count (r = 0.471; p = 0.001). No significant correlation was encountered between EMPs and hematological and biochemical markers, including serum ferritin or D-dimer levels.
Table 2: Assessment of correlations between levels of erythrocyte-derived micro-particles (EMPs), platelets-derived micro-particles (PMPs), and N-Terminal pro-B-type natriuretic peptide (NT-proBNP) with other variables in children with NTDT (N = 50).
|
EMPs vs.
|
|
Hemoglobin level, g/dL
|
0.140
|
0.333
|
|
Red blood cell count, ×106/UL
|
0.202
|
0.160
|
|
Reticulocytes, %
|
-0.228
|
0.111
|
|
Lactate dehydrogenase, U/L
|
0.134
|
0.353
|
|
Ferritin, ng/mL
|
0.067
|
0.645
|
|
D-dimer, µg/L
|
-0.024
|
0.866
|
|
PMPs vs.
|
|
Platelets count, ×103/µL
|
0.471
|
0.001*
|
|
D-dimer, µg/L
|
0.121
|
0.402
|
|
Ferritin, ng/mL
|
0.226
|
0.114
|
|
NT-proBNP level vs.
|
|
Hemoglobin level, g/dL
|
0.006
|
1.000
|
|
Platelets count, ×103/µL
|
0.080
|
0.582
|
|
D-dimer level, µg/L
|
0.137
|
0.344
|
|
Ferritin level, µg/mL
|
0.051
|
0.724
|
|
EMPs, %
|
0.098
|
0.499
|
NTDT: non-transfusion-dependent thalassemia; r: Pearson’s correlation coefficient; *Significant.
Based on D-dimer levels, NTDT patients were categorized into two subgroups: normal (< 550 µg/L, n = 29) and high (≥ 550 µg/L, n = 21). No significant differences were observed between these subgroups in Hb, RBC count, WBC count, reticulocytes, platelet count, or thrombocytosis prevalence [Table 3]. Similarly, there were no significant differences in PMP or EMPs levels. However, patients with high D-dimer had significantly higher levels of serum ferritin (p = 0.012), NT-ProBNP (p = 0.047), and lactate dehydrogenase (p = 0.032) [Table 3].
Table 3: Hematological characteristics, microparticles, serum ferritin, and NTproBNP levels of NTDT patients compared based on D-Ddimer level (hypercoagulable status) (N=50).
|
Hemoglobin, g/dL
|
|
Min–Max
|
5.7–9.3
|
5.2–11.9
|
t(df=48) = 0.517
p = 0.608
|
|
Mean ± SD
|
7.6 ± 1.0
|
7.4 ± 1.4
|
|
Red blood cell count, ×106/μL
|
|
Min–Max
|
2.4–4.9
|
2.8–5.7
|
t(df=48) =1.037
p = 0.305
|
|
Mean ± SD
|
3.7 ± 0.6
|
3.90 ± 0.7
|
|
Reticulocytes, %
|
|
Min–Max
|
1.5–6.8
|
0.5–6.8
|
t(df=48) =1.478
p=0.146
|
|
Mean ± SD
|
3.5 ± 1.3
|
4.1 ± 1.5
|
|
White blood cell count, ×109/L
|
|
Min–Max
|
2.8–18.0
|
4.6–31.5
|
t(df=48) = 1.729
p = 0.090
|
|
Mean ± SD
|
8.0 ± 3.1
|
10.6 ± 7.2
|
|
Platelets count, ×106/μL
|
|
Min–Max
|
153.0–807.0
|
146.0–1153.0
|
t(df=48) = 0.149
p = 0.882
|
|
Mean ± SD
|
334.0 ± 154.6
|
325.0 ± 268.6
|
|
Thrombo-cytosis, n (%)
|
5 (17.2)
|
4 (19.1)
|
Z = 0.164
p = 0.873 NS
|
|
Lactate dehydrogenase, U/L
|
|
Min-Max
|
164.0–410.0
|
190.0–420.0
|
t(df=48) = 2.251
p = 0.032*
|
|
Mean ± SD
|
241.5 ± 68.7
|
287.8 ± 75.8
|
|
Platelets-derived micro-particles, %
|
|
Min–Max
|
1.0–14.0
|
2.0–28.1
|
t(df=48) = 0.925
p = 0.0360
|
|
Mean ± SD
|
5.6 ± 3.2
|
6.8 ± 5.7
|
|
Erythrocyte-derived micro-particles, %
|
|
Min–Max
|
0.3–20.0
|
0.5–20.0
|
t(df=48) = 0.660
p = 0.512
|
|
Mean ± SD
|
4.1 ± 4.1
|
5.0 ± 5.4
|
|
Serum Ferritin, µg/L
|
|
Min–Max
|
17.9–1700.0
|
56.0–2086.0
|
t(df=48) = 2.604
p = 0.012*
|
|
Mean ± SD
|
312.8 ± 336.2
|
619.0 ± 495.9
|
|
N-Terminal pro-B-type natriuretic peptide, pg/mL
|
|
Min–Max
|
64.5–123.2
|
75.3–142.0
|
NTDT: non-transfusion-dependent thalassemia; t: Student’s t-test; Z: Z test for comparison of two independent proportions; df: degree of freedom; * Significant.
In the present study, serum ferritin level was a statistically significant discriminator of hypercoagulability (D-dimer ≥ 550 µg/L) with an area under the receive operator characteristics (ROC) curve (AUC) of 0.740 (95% CI: 0.596–0.853; Z = 3.305; p = 0.001). The optimal cutoff value determined using the Youden index was > 277.3 µg/L with a sensitivity of 81.0% (95% CI: 58.09–94.55), specificity of 69.0% (95% CI: 49.17–84.72), positive predictive value of 65.4% (95% CI: 51.38–77.15), Negative Predictive Value of of 83.3% (95% CI: 66.70–92.58). The overall accuracy was 74.0% (95% CI: 59.7–85.4; p = 0.014). The odds ratio of hypercoagulability at serum ferritin levels > 277.3 µg/L was 9.400 (95% CI: 2.464–36.198).
A predictive model for hypercoagulability (D-dimer ≥ 550 µg/L) was developed with PMPs, EMPs, and ferritin as independent variables. The model was tested by using AUC (naive Bayes method), which yielded 71.1%, with a classification accuracy of 68.0% and a precision of 63.2%. A Monte Carlo data simulation on a hypothetical sample of 250 patients (derived from the current study data) improved the area under ROC curve to 100%, with classification accuracy and precision both reaching 98.4%. This suggests that our model predict hypercoagulability in larger patient samples [Figure 2].
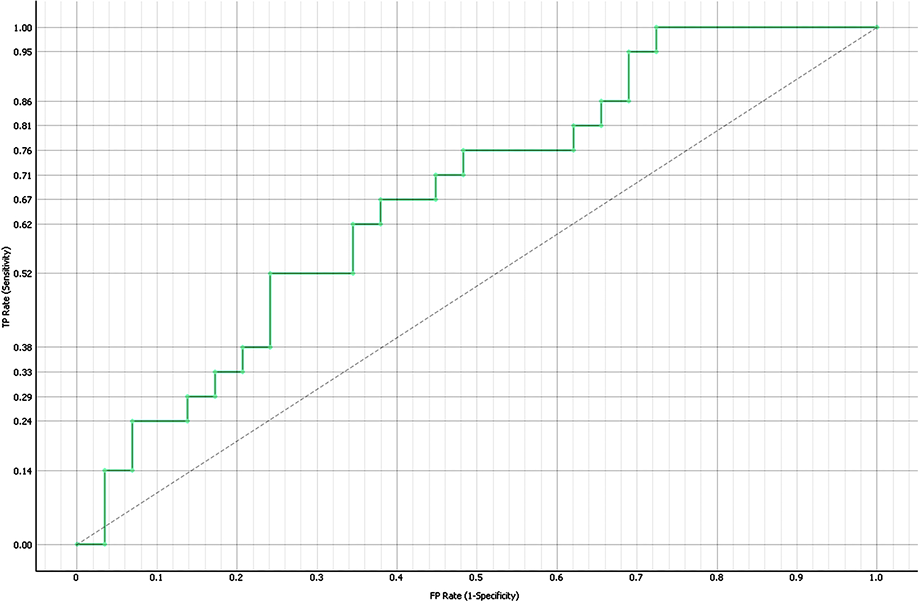 Figure 2: Receiver operating characteristic (ROC) curve of the logistic regression model’s ability to discriminate hypercoagulability (D-dimer ≥ 550 µg/L) from non-hypercoagulability. Each point on the green stepped curve illustrates the discriminative performance of the model based on combined predictors, with the diagonal dotted line representing no discriminative ability. The y-axis shows sensitivity (true-positive rate) and the x-axis shows specificity (false-positive rate.
Figure 2: Receiver operating characteristic (ROC) curve of the logistic regression model’s ability to discriminate hypercoagulability (D-dimer ≥ 550 µg/L) from non-hypercoagulability. Each point on the green stepped curve illustrates the discriminative performance of the model based on combined predictors, with the diagonal dotted line representing no discriminative ability. The y-axis shows sensitivity (true-positive rate) and the x-axis shows specificity (false-positive rate.
Discussion
Patients with β-thalassemia have a higher risk for hypercoagulability and thrombosis, with potential for morbidity and mortality in the long term. This phenomenon involves interactions between damaged RBCs, activated leukocytes and platelets, adhesive endothelial cells, and coagulation factor dysregulation.11,21
Circulating cell-derived vesicles including MPs tend to be elevated in thalassemia, especially in splenectomized patients. Several studies have revealed a notable presence of plasma MPs primarily released from platelets and damaged red cells which express negatively charged phospholipids.7,22,23 In patients with thalassemia, various studies revealed a notable presence of plasma MPs, which are primarily derived from platelets and damaged RBCs, which promote platelet aggregation, contributing to thrombus formation.7
The present study evaluated EMPs and PMPs in 50 children with NTDT and 50 matched healthy controls. EMPs were significantly higher in patients than in controls, suggesting a potential role in initiation of hypercoagulability. Our finding aligns with several previous reports.4,5,24-26
We could not elicit significant correlations between EMP levels and variables such as Hb level, RBC count, reticulocyte count, LDH, or serum ferritin. However, in Egyptian pediatric patients with thalassemia intermedia, a previous study found a significant positive correlation between EMP and ferritin levels. Further, EMP had a significant negative correlation with Hb level but none with RBC count or LDH level.24 In mixed-age cohorts of urban Egyptian patients, previous studies reported a negative correlation between EMP levels and hemoglobin levels.4,25 Variations in sample size, age distribution, and clinical characteristics may explain the differences in correlation findings.
PMPs are procoagulant subcellular vesicles released from activated platelets and are considered biomarkers for thromboembolic risk in both thalassemia major (TM) and intermedia (TI), and in our cohort, the PMP levels were significantly higher among NTDT patients than in controls, consistent with other studies.4,5,9,23, 26–28,29 We also found a significant positive correlation between PMPs and platelet count, suggesting chronic platelet activation. Meanwhile, a 2022 study among a mixed-age cohort in Assiut, Egypt, reported a significantly positive correlation between PMP level and platelet count in splenectomized β-TI patients, but not in the overall cohort.29
Enhanced platelet aggregation and increased platelet activation markers reflect systemic platelet activation.30 In the present study, platelet aggregation remained within the normal range in all patients (75%–85%).23 Similarly, in Iran, normal platelet aggregation (80.4 ± 9.4) was reported among 36 young adults with β-TI; the authors hypothesized that chronic platelet activation in β-TI may render the activated platelets refractory to further stimulation.31 Literature on platelet aggregation in thalassemia is mixed, with reports of increased,22,23 decreased,32 or normal reactivity,10,33 attributable to differences in laboratory techniques, disease characteristics, or racial/regional variations.33
D-dimer level serves as an indirect marker for thrombotic risk and fibrinolysis.29 In our study, the mean D-dimer level exceeded the normal threshold of < 550 µg/L for 21 (42.0%) patients, five (24%) of whom were splenectomized. An extensive regional clinical study in 2008, covering Mediterranean countries (including Egypt) and Iran, analyzed data from 8860 thalassemia patients (6670 TM and 2190 TI) and identified splenectomy and age over 20 years as the main risk factors for thrombosis.34 None of our patients had thrombotic manifestations, likely due to their younger age, as thrombotic events typically manifest after age 18.
Our study found serum ferritin level to be a good discriminator of hypercoagulability, with good sensitivity, specificity, and overall accuracy. The odds of hypercoagulability above this threshold were 9.4 times higher. Youssry et al. (2017)4 also reported significantly higher ferritin levels in patients with thromboembolic events. In a large cohort study of 584 patients with thalassemia intermedia from Mediterranean countries and Iran, serum ferritin levels ≥1000 ng/L and age > 20 years were significantly associated with increase risk of thrombosis.35
We observed no significant difference in hematological indices or microparticle percentages between patients with normal and high D-dimer possibly due to sample size limitations. However, we developed a nomogram with high performance based on the three key predictors of hypercoagulability (Ferritin, EMPs, PMPs) as a simple, noninvasive tool for early prediction of thrombotic risk in NTDT patients.
NT-proBNP has been proposed as a non-invasive biomarker for the early detection of PH and right ventricular strain. In patients with NTDT, elevated NT-proBNP levels may reflect subclinical cardiopulmonary involvement and thromboembolic risk.36 Recently, measurement of serum level of NT-proBNP has been proposed as a non-invasive biomarker for early detection of PHT.37,38
In the present study, the mean proBNP level was higher in NTDT patients but without statistical significance. Several other studies also reported significantly higher NT-proBNP levels.39–41 However, these studies were in adult thalassemic patients with TM and TI. An earlier study reported significantly elevated NT-ProBNP levels in patients with PHT. However, even patients without PHT had high levels compared with controls, suggesting that NT-ProBNP may be a strong indicator of PH. 14
Although we did not find a direct correlation between NT-proBNP and D-dimer, our patients with elevated D-dimer levels had significantly higher NT-proBNP levels. This suggests that the occurrence of microthrombi in pulmonary circulation may lead to right ventricular strain and increased release of NT-proBNP.
The present study has limitations, including the relatively small sample size, and being conducted at a single center, limiting its generalizability, calling for larger multicenter studies with longer follow-up periods.
Conclusion
No overt thrombotic events were detected in this cohort of Egyptian children with NTDT. However, elevated D-dimer levels may serve as indirect markers of subclinical thrombotic risk and ongoing fibrinolysis, highlighting a hypercoagulable state even in the absence of clinically evident thrombosis. Assessment of endothelial and PMPs may provide additional value for early detection of hypercoagulability and thrombotic risk. Furthermore, measurement of NT-proBNP may be useful as a non-invasive screening tool for early cardiopulmonary involvement, including PH, particularly in high-risk patients. Larger multicenter studies with longer follow-up are warranted to validate these findings and to establish evidence-based strategies for risk stratification and management.
Disclosure
The authors declare no conflicts of interest. No funding was received for this study.
references
- 1. Musallam KM, Rivella S, Vichinsky E, Rachmilewitz EA. Non-transfusion-dependent thalassemias. Haematologica 2013 Jun;98(6):833-844.
- 2. Rameli N, Ramli M, Zulkafli Z, Hassan MN, Yusoff SM, Noor NH, et al. Challenges in the diagnosis of beta-thalassemia syndrome: the importance of molecular diagnosis. Oman Med J 2022 Jan;37(1):e331.
- 3. Farmakis D, Porter J, Taher A, Domenica Cappellini M, Angastiniotis M, Eleftheriou A. 2021 thalassaemia international federation guidelines for the management of transfusion-dependent thalassemia. Hemasphere 2022 Jul;6(8):e732.
- 4. Youssry I, Soliman N, Ghamrawy M, Samy RM, Nasr A, Abdel Mohsen M, et al. Circulating microparticles and the risk of thromboembolic events in Egyptian beta thalassemia patients. Ann Hematol 2017 Apr;96(4):597-603.
- 5. Habib A, Kunzelmann C, Shamseddeen W, Zobairi F, Freyssinet JM, Taher A. Elevated levels of circulating procoagulant microparticles in patients with beta-thalassemia intermedia. Haematologica 2008 Jun;93(6):941-942.
- 6. Jiskani SA. Extracellular vesicles in thalassemia: mechanisms, implications, and therapeutic potential. Asp Mol Med 2025;5:100061.
- 7. Klaihmon P, Pattanapanyasat K, Phannasil P. An update on recent studies of extracellular vesicles and their role in hypercoagulability in thalassemia (review). Biomed Rep 2023 Dec;20(2):31.
- 8. Shash H. Non-transfusion-dependent thalassemia: a panoramic review. Medicina (Kaunas) 2022 Oct;58(10):1496.
- 9. Cappellini MD, Motta I, Musallam KM, Taher AT. Redefining thalassemia as a hypercoagulable state. Ann N Y Acad Sci 2010 Aug;1202(1):231-236.
- 10. Atichartakarn V, Chuncharunee S, Chandanamattha P, Likittanasombat K, Aryurachai K. Correction of hypercoagulability and amelioration of pulmonary arterial hypertension by chronic blood transfusion in an asplenic hemoglobin E/beta-thalassemia patient. Blood 2004 Apr;103(7):2844-2846.
- 11. Klaihmon P, Phongpao K, Kheansaard W, Noulsri E, Khuhapinant A, Fucharoen S, et al. Microparticles from splenectomized β-thalassemia/HbE patients play roles on procoagulant activities with thrombotic potential. Ann Hematol 2017 Feb;96(2):189-198.
- 12. Matusov Y, Kolaitis NA, Geft D, DesJardin J, Barnett C, Hage A, et al. How I do it: best practices for right heart catheterization in the diagnosis of pulmonary hypertension. Chest 2025 May;S0012-3692(25)00579-3.
- 13. Fraidenburg DR, Machado RF. Pulmonary hypertension associated with thalassemia syndromes. Ann N Y Acad Sci 2016 Mar;1368(1):127-139.
- 14. Voskaridou E, Tsetsos G, Tsoutsias A, Spyropoulou E, Christoulas D, Terpos E. Pulmonary hypertension in patients with sickle cell/beta thalassemia: incidence and correlation with serum N-terminal pro-brain natriuretic peptide concentrations. Haematologica 2007 Jun;92(6):738-743.
- 15. Taher AT, Musallam KM, Cappellini MD. Guidelines for the management of non-transfusion-dependent β-thalassaemia. Nicosia (Cyprus): Thalassaemia International Federation; 2023.
- 16. Chopra N, Doddamreddy P, Grewal H, Kumar PC. An elevated D-dimer value: a burden on our patients and hospitals. Int J Gen Med 2012;5:87-92.
- 17. Elsayh KI, Zahran AM, El-Abaseri TB, Mohamed AO, El-Metwally TH. Hypoxia biomarkers, oxidative stress, and circulating microparticles in pediatric patients with thalassemia in Upper Egypt. Clin Appl Thromb Hemost 2014 Jul;20(5):536-545.
- 18. Field A. Discovering statistics using IBM SPSS statistics. 4th ed. London, California, New Delhi: SAGE Publications Ltd; 2013.
- 19. Demšar J, Curk T, Erjavec A, Gorup Č, Hočevar T, Milutinovič M, et al. Orange: data mining toolbox in Python. Journal of machine Learning research 2013;14(1):2349-2353.
- 20. Curran-Everett D. Evolution in statistics: p values, statistical significance, kayaks, and walking trees. Adv Physiol Educ 2020 Jun 1;44(2):221-224.
- 21. Cappellini MD, Musallam KM, Poggiali E, Taher AT. Hypercoagulability in non-transfusion-dependent thalassemia. Blood Rev 2012 Apr;26(Suppl 1):S20-S23.
- 22. Tantawy AA, Adly AA, Ismail EA, Habeeb NM. Flow cytometric assessment of circulating platelet and erythrocytes microparticles in young thalassemia major patients: relation to pulmonary hypertension and aortic wall stiffness. Eur J Haematol 2013 Jun;90(6):508-518.
- 23. Bhattacharyya M, Kannan M, Chaudhry VP, Mahapatra M, Pati H, Saxena R. Hypercoagulable state in five thalassemia intermedia patients. Clin Appl Thromb Hemost 2007 Oct;13(4):422-427.
- 24. Shahin RS, Al-Habibi A-S, Raga Abdel-Salam M, Gouda RM. Circulating erythrocyte derived microparticles in pediatric thalassemia intermedia patients. The Journal of the Egyptian Society of Haematology & Research 2005;13(1):1.
- 25. Mohammed Mowafy N, Zaki Ali El Zohairy Y, Mahmoud Hussien S, Saleh Sadek Mohamed A. Evaluation of red blood cell microparticles in thalassemia. Azhar Med J 2016;45(3):611-620.
- 26. Chinsuwan J, Klaihmon P, Kadegasem P, Chuansumrit A, Soisamrong A, Pattanapanyasat K, et al. High prevalence of antiphospholipid antibodies in children with non-transfusion dependent thalassemia and possible correlations with microparticles. Mediterr J Hematol Infect Dis 2020 Nov;12(1):e2020071.
- 27. Moawad MR, Eldash HH, Hussein SK, Eid MM. Platelet derived micro particles and the risk of pulmonary hypertension in Egyptian patients with thalassemia major. Life Sci J 2022;19(12):30-35.
- 28. Sirachainan N. Thalassemia and the hypercoagulable state. Thromb Res 2013;132(6):637-641.
- 29. Abdelaziz HM, El-Beih EA, Sayed DM, Afifi OA, Thabet AF, Elgammal S, et al. Increased levels of circulating platelet microparticles as a risk of hypercoagulable state in β-thalassemia intermedia patients. The Egyptian Journal of Haematology 2022;47(3):187-193.
- 30. Abo-Elwafa HA, Youseff LM, Mahmoud RA, Elbadry MI, Tawfeek A, Aziz SP. Venous thromboembolism risk assessment among beta-thalassemia patients. Journal of Applied Hematology 2023;14(3):230-235.
- 31. Zahedpanah M, Azarkeivan A, Ahmadinejad M, Tabatabaiee M, Hajibeigi B, Maghsudlu M. Evaluation of platelet aggregation in splenectomized beta-thalassemia major and intermedia patients. Journal of Applied Hematology 2018;9(4):126-130.
- 32. Chaudhary H, Ahmad N. Frequency of platelet aggregation defects in children suffering fromg β-thalassemia. Saudi Journal for Health Sciences 2012;1(2):92-98.
- 33. Eldor A, Rachmilewitz EA. The hypercoagulable state in thalassemia. Blood 2002 Jan;99(1):36-43.
- 34. Taher A, Isma’eel H, Mehio G, Bignamini D, Kattamis A, Rachmilewitz EA, et al. Prevalence of thromboembolic events among 8,860 patients with thalassaemia major and intermedia in the Mediterranean area and Iran. Thromb Haemost 2006 Oct;96(4):488-491.
- 35. Taher AT, Musallam KM, Karimi M, El-Beshlawy A, Belhoul K, Daar S, et al. Overview on practices in thalassemia intermedia management aiming for lowering complication rates across a region of endemicity: the optimal care study. Blood 2010 Mar 11;115(10):1886-1892.
- 36. Sleiman J, Tarhini A, Bou-Fakhredin R, Saliba AN, Cappellini MD, Taher AT. Non-transfusion-dependent thalassemia: an update on complications and management. Int J Mol Sci 2018 Jan;19(1):182.
- 37. Yap LB, Ashrafian H, Mukerjee D, Coghlan JG, Timms PM. The natriuretic peptides and their role in disorders of right heart dysfunction and pulmonary hypertension. Clin Biochem 2004 Oct;37(10):847-856.
- 38. Gupta V, Vijayakumar V, Aggarwal P, Kumar I, Agrawal V. Pulmonary artery hypertension in transfusion-dependent thalassemia. Indian Pediatr 2024 Jan;61(1):49-52.
- 39. Safniyat S, Shakibazad N, Haghpanah S, Amoozegar H, Karimi M, Safaei S, et al. Parameters of tissue iron overload and cardiac function in patients with thalassemia major and intermedia. Acta Haematol Pol 2020;51(2):95-101.
- 40. Mohamed H, El Zimaity M, Abdelbary H. Brain natriuretic peptide as a sensitive biomarker for early detection of cardiac affection in adult Egyptian patients with β-thalassemia. The Egyptian Journal of Haematology 2019;44(1):34-39.
- 41. Balkan C, Tuluce SY, Basol G, Tuluce K, Ay Y, Karapinar DY, et al. Relation between NT-proBNP levels, iron overload, and early stage of myocardial dysfunction in β-thalassemia major patients. Echocardiography 2012 Mar;29(3):318-325.