Pulmonary arterial hypertension (PAH) is a progressive disorder marked by elevated pulmonary artery pressure, leading to right heart failure and significant morbidity.1 The pathophysiology of PAH is intricate, involving a multitude of factors that result in increased pulmonary vascular resistance and subsequent cardiac complications.2 Current therapeutic strategies aim to mitigate these hemodynamic disturbances, yet they vary in effectiveness and are often accompanied by serious complications such as pericardial effusion, which can further exacerbate patient outcomes.3,4
The prognostic implications of pericardial effusion in PAH patients are complex. While effusions can indicate disease severity and worsened prognosis,5,6 the decision to perform pericardiocentesis must be weighed carefully against the risks.3 This study aims to evaluate the impact of various PAH therapies on the incidence of pericardial effusion and patient survival, exploring whether treatment modalities can alter the course of this complication and improve long-term outcomes. Through this investigation, we seek to add to the body of evidence that pericardial effusion is a negative prognostic marker in PAH and to examine the dire conjunction of severe PAH with pericardial effusion.7,8
Methods
Our retrospective cohort study methodically enrolled patients diagnosed with PAH at a high-volume tertiary care center in the USA, from January 2015 and December 2019. These patients started treatment with intravenous (IV) epoprostenol or IV/ subcutaneous (SC) treprostinil as part of their management plan.
The study encompassed a comprehensive review of patient data including: 1) echocardiographic assessments for cardiac structure and function; 2) right heart catheterization for direct hemodynamic measurements; 3) World Health Organization functional class evaluations to determine disease severity; 4) six-minute walk distance tests to assess exercise capacity; and 5) brain natriuretic peptide (BNP) levels as a biomarker for cardiac strain. Additional markers and tests were also considered, including: 1) N-terminal prohormone of brain natriuretic peptide (NT-proBNP) as an alternative to BNP for assessing cardiac stress; 2) cardiac magnetic resonance imaging for detailed structural and functional analysis, when available; and 3) serum markers of inflammation (e.g., C-reactive protein) and cardiac injury (e.g., troponins) that could correlate with disease progression and treatment response.
The timing of these evaluations was at the attending cardiologist’s discretion. A significant follow-up point was defined by the first post-treatment echocardiogram, which occurred a minimum of 90 days after starting IV/SC therapy. Survival outcomes were correlated with baseline and first follow-up hemodynamic, echocardiographic parameters, and biochemical markers, analyzing the impact of parenteral prostanoid therapy.
Inclusion criteria included: patients diagnosed with PAH (as per the European Society of Cardiology and the European Respiratory Society guidelines) between January 2015 and December 2019; aged ≥ 18 years; and had undergone at least one form of PAH-specific therapy, including but not limited to, endothelin receptor antagonists, phosphodiesterase type 5 inhibitors, or prostacyclin analogs.
Patients with secondary causes of pulmonary hypertension such as chronic thromboembolic pulmonary hypertension, left heart disease, lung diseases, or chronic hypoxia were excluded in the study as well as history of pericardial disease before PAH diagnosis, and incomplete medical records or insufficient follow-up data.
We adopted a retrospective cohort study design, utilizing patient data extracted from electronic medical records. Ethical approval was obtained from the institutional review board, and all procedures were conducted in accordance with the Declaration of Helsinki.
The analysis focused on evaluating the efficacy and safety of PAH therapies, with a particular interest in the development of pericardial effusion as a potential side effect and its impact on patient survival. Statistical analyses were performed using SPSS (IBM Corp. Released 2017. IBM SPSS Statistics for Windows, Version 25.0. Armonk, NY: IBM Corp.). A p-value of < 0.05 was considered statistically significant.
Results
In our comprehensive longitudinal study, we assessed a cohort of 60 patients diagnosed with PAH, following the initiation IV/SC therapy over a median observation period of four years. Our cohort predominantly consisted of male patients, accounting for 62.0% with a median age of 59 years at the initiation of therapy [Table 1]. The interim between the PAH diagnosis and the commencement of therapy averaged approximately 391 days, suggesting potential delays in the treatment onset.
Table 1: Major difference in etiology, hemodynamics, echo findings, and effusion before and after therapy.
|
Patients, n
|
41
|
19
|
|
Idiopathic PAH
|
25
|
12
|
|
CTD PAH
|
10
|
5
|
|
CHD PAH
|
6
|
2
|
|
Log NT-proBNP
|
3.1
|
3.5
|
|
6MWD, feet
|
350
|
300
|
|
RA
|
12
|
18
|
|
Mean PA, mmHg
|
55.0
|
60.0
|
|
PCWP, mmHg
|
9
|
12
|
|
PVR, Wood units
|
2
|
4
|
|
CI, L/min/m2
|
2.3
|
2.0
|
|
RA size normal-mild enlargement
|
30
|
8
|
|
RA size moderate enlargement
|
10
|
10
|
|
RA size severe enlargement
|
1
|
1
|
|
LA size normal-mild enlargement
|
35
|
15
|
|
LA size moderate enlargement
|
5
|
3
|
|
LA size severe enlargement
|
1
|
1
|
|
RV size normal-mild enlargement
|
25
|
10
|
|
RV size moderate enlargement
|
15
|
7
|
|
RV size severe enlargement
|
1
|
2
|
|
RV function normal-mild dysfunction
|
30
|
10
|
|
RV function moderate dysfunction
|
10
|
5
|
|
RV function severe dysfunction
|
1
|
4
|
|
TR severity none-mild
|
35
|
5
|
|
TR severity moderate
|
5
|
10
|
|
TR severity severe
|
1
|
4
|
|
IVC size normal
|
30
|
7
|
|
IVC size dilated
|
10
|
9
|
PAH: pulmonary arterial hypertension; CTD: connective tissue disease;
CHD: congenital heart disease; NT-proBNP: N-terminal prohormone of brain natriuretic peptide; 6MWD: six-minute walk distance; RA: right atrium; PA: pulmonary artery pressure; PCWP: pulmonary capillary wedge pressure; PVR: pulmonary vascular resistance; CI: cardiac index; LA: left atrium;
RV: right ventricle; TR: tricuspid regurgitation; IVC: inferior vena cava.
Throughout the observation period, the survival rates for patients showed a high initial one-year survival rate of 89.0%. However, there was a notable decline in survival to 71.0% by the end of the third year post-therapy initiation. This decline signals a potentially critical window wherein enhanced patient management and timely intervention might be pivotal.
Echocardiographic assessments conducted before the initiation of therapy indicated the presence of pericardial effusion in 31.7% of the patients. Notably, the etiology of PAH showed no significant differences between patients with or without pericardial effusion, suggesting that the occurrence of effusion is independent of the underlying cause of PAH. Nevertheless, the presence of pericardial effusion was markedly correlated with more severe manifestations of PAH. This was evidenced by the elevated levels of log BNP, increased right atrial pressures, along pronounced tricuspid regurgitation and inferior vena cava enlargement [Figure 1]. These markers are indicative of heightened cardiovascular stress among PAH patients.

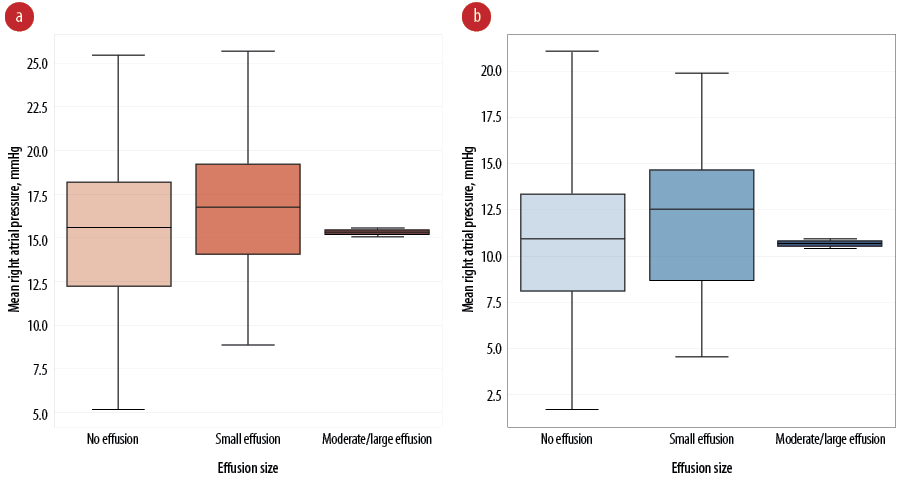 Figure 1: Comparative analysis of pericardial effusion size and right atrial pressure (a) before and (b) after therapeutic intervention.
Figure 1: Comparative analysis of pericardial effusion size and right atrial pressure (a) before and (b) after therapeutic intervention.
A profound finding from our study was the differential impact of pericardial effusion size on patient outcomes. Moderate to large effusions, though present in a small subset of only two patients (3.3%), exhibited a significant increase in mortality risk, with a hazard ratio (HR) of 1.92 and a 95% CI ranging from 1.10 to 44.78 (p = 0.004). Conversely, small pericardial effusions appeared to confer a protective effect (HR = 0.27, 95% CI: 0.15–0.48; p = 0.006). The mortality rate in the early post-therapy phase was significantly elevated in patients presenting with any degree of pericardial effusion, with the effect being more pronounced in those with larger effusions.
Our findings underscore the heterogeneity of pericardial effusion implications in PAH and highlight the necessity for a stratified approach in patient care. The correlation between effusion size and patient prognosis provides a compelling direction for future research, to refine therapeutic strategies to improve long-term outcomes in PAH patients.
Discussion
In this study, we observed a cohort of 60 PAH patients under IV/SC therapy, revealing significant insights into the prognosis and management of the disease, particularly about pericardial effusion. Our findings, including the survival rates and the association of pericardial effusion with mortality, offer valuable contributions to the existing literature on PAH management.
Our observation that survival rates declined from 89.0% after one year to 71.0% by the third year is consistent with previous studies indicating a critical need for enhanced monitoring and possibly early intervention strategies in PAH patients.2 The prevalence and prognostic significance of pericardial effusion in our cohort align with previous studies, underscoring the complexity of managing advanced PAH.4,5
Notably, our study diverges from De Filippo et al,7 regarding the prognostic implications of small effusions, which we found to be inversely correlated with mortality risk. This discrepancy highlights the potential for small effusions to serve as markers for less advanced disease stages or to possess a protective physiological mechanism, a novel insight that warrants further investigation.
A major strength of our study is the detailed longitudinal follow-up of patients, allowing for a nuanced understanding of the disease progression and therapy outcomes over time. The focus on the association between pericardial effusion size and patient survival further contributes to the literature by clarifying the prognostic value of effusion characteristics in PAH management.
However, our study has some limitations. The predominantly male cohort may limit the generalizability of our findings to the broader PAH patient population, as gender differences in PAH pathophysiology and outcomes have been documented.2 Additionally, the observational nature of our study precludes definitive conclusions about causality between pericardial effusion and mortality.
Future studies should aim to elucidate the pathophysiological underpinnings of pericardial effusion in PAH and explore the impact of targeted interventions on patient outcomes. Prospective research could also investigate the potential protective role of small effusions and the mechanisms underlying their inverse correlation with mortality risk.
Conclusion
Our study elucidates the prognostic value of pericardial effusion in PAH, revealing that a moderate to large effusion at the onset of parenteral prostanoid therapy heralds a higher mortality risk. This finding accentuates the critical role of vigilant monitoring and timely intervention for pericardial effusion in PAH patients. It advocates for an integrated approach within the therapeutic protocol, suggesting that management of pericardial effusion could be pivotal in improving patient prognosis. Future therapeutic strategies should incorporate the severity of pericardial effusion as a key consideration in treatment planning.
Disclosure
The authors declared no conflicts of interest. No funding was received for this study.
references
- 1. Cassady SJ, Ramani GV. Right heart failure in pulmonary hypertension. Cardiol Clin 2020 May;38(2):243-255.
- 2. Strange G, Stewart S, Celermajer DS, Prior D, Scalia GM, Marwick TH, et al; NEDA Contributing Sites. NEDA Contributing Sites. Threshold of pulmonary hypertension associated with increased mortality. J Am Coll Cardiol 2019 Jun;73(21):2660-2672.
- 3. Shadrin IY, Idossa D, Walston V, Lee J III, Issa M. Pulmonary hypertension and pericardial effusions-to tap or not to tap. Am J Med 2020 Apr;133(4):e149-e150.
- 4. Fenstad ER, Le RJ, Sinak LJ, Maradit-Kremers H, Ammash NM, Ayalew AM, et al. Pericardial effusions in pulmonary arterial hypertension: characteristics, prognosis, and role of drainage. Chest 2013 Nov;144(5):1530-1538.
- 5. Batal O, Dardari Z, Costabile C, Gorcsan J, Arena VC, Mathier MA. Prognostic value of pericardial effusion on serial echocardiograms in pulmonary arterial hypertension. Echocardiography 2015 Oct;32(10):1471-1476.
- 6. Abu-Rmaileh M, Mirza O, Patel C, Shah T, Hardin EA, Bartolome SD, et al. Prognosis of pulmonary arterial hypertension patients with pericardial effusion before and after initiation of parenteral prostacyclin therapy. Pulm Circ 2023 Apr;13(2):e12226.
- 7. De Filippo O, Gatti P, Rettegno S, Iannaccone M, D’Ascenzo F, Lazaros G, et al. Is pericardial effusion a negative prognostic marker? Meta-analysis of outcomes of pericardial effusion. J Cardiovasc Med (Hagerstown) 2019 Jan;20(1):39-45.
- 8. Pahuja M, Weeratunga A, Feldman J. Severe pulmonary artery hypertension (PAH) and pericardial effusion (PE): a deadly combination: a case series and review of literature. Chest 2016;150(4):1168A.