Acute testicular pain with or without scrotal swelling (referred to as ‘acute scrotum’) is a common cause of referral to the emergency department (ED). The symptoms may be accompanied by local signs or general symptoms. All patients with acute scrotum have to be triaged with high priority in EDs, as the causes of the symptoms may be serious (e.g., testicular torsion or strangulated hernia) and require emergency surgical intervention.1,2 Other differential diagnoses for acute scrotum are torsion of the testicular appendix, epididymitis, epididymo-orchitis, symptomatic spermatocele/hydrocele/varicocele, scrotal abscess including scrotal furunculosis, testicular trauma, distal ureterolithiasis, and testicular cancer.3–5
Color Doppler ultrasound does not provide a reliable diagnosis of acute testicular torsion and can therefore be misleading and delay appropriate treatment (surgical exploration).6 There is a recent report in a small number of patients on the promising use of transscrotal near infrared spectroscopy in the evaluation of adult acute scrotum.7 To date, however, no tool or examination can reliably distinguish emergency situations such as testicular torsion from less acute causes of scrotal pain such as epididymitis, spermatocele, hydrocele, or distal ureterolithiasis. We, therefore, conducted a large observational study of 440 patients presenting with acute scrotum to our ED in an attempt to establish a decision tree that can facilitate timely and effective treatment of patients with acute scrotum.
Methods
We conducted a retrospective analysis of 440 consecutive patients referred with acute scrotum to the ED of the University Hospital of Bern, Switzerland, between January 2003 and January 2013. The following patient characteristics were assessed: time between onset of pain and arrival at the ED, characteristics and localization of pain, and Prehn’s sign (positive or negative). All patients underwent clinical/physical, laboratory (blood analysis, urinalysis), and ultrasound examinations. Blood C-reactive protein (CRP) > 3 mg/L, total blood leucocytes (WBC) count > 10.4 g/L, and microscopically > 4 WBC or > 4 erythrocytes (Ec) per field were considered pathological. After the first assessment of the patients by emergency doctors, our hospital urologists including one of four senior urologists were involved in the care of all patients and made subsequent decisions on treatment (hospitalization, surgery, medical treatment, discharge).
To assess factors that help to distinguish between genital/paragenital infection and testicular torsion, simple and multiple binary logistic regression analyses were performed on the different laboratory parameters (CRP </> 3 ml/L, WBC </> 10.4 g/L, </> 4 WBC per field in urine, </> 4 Ec per field in urine), time between onset of pain and arrival in the ED, characteristics of pain, localization of pain, and Prehn’s sign (positive or negative). This provided odds ratios (OR) with 95% confidence intervals (95% CI) and the corresponding p-value for each variable. All p-values and OR (95% CI) were based on the Wald test. All analyses were performed using R version 2.9.2 (R Foundation for Statistical Computing, Vienna, Austria).
This study was conducted according to local law. As only anonymous data was used in this study, no patient consent nor an ethical permission was needed.
Results
The median age of our cohort was 30 years (range: 16–76). Ninety-one percent (404) of patients were referred with testicular pain of ≤ 3 days duration, while 8.7% (36) had complained of testicular pain for > 3 days. Most patients presented with genital/paragenital infections (58.4%; 257) or chronic testicular pain (11.8%; 52). Testicular torsion was diagnosed in 9.5% (42) of patients, testicular cancer in 4.3% (19), and symptomatic distal ureterolithiasis in 2.5% (11) of patients [Table 1].
Table 1: Final diagnosis in patients referred to the emergency department with acute scrotum.
|
Genital/paragenital infection |
257 (58.4) |
|
Chronic testicular pain |
52 (11.8) |
|
Testicular torsion |
42 (9.5) |
|
Testicular trauma |
22 (5.0) |
|
Testicular cancer |
19 (4.3) |
|
Symptomatic distal ureterolithiasis |
11 (2.5) |
|
Symptomatic spermatocele |
7 (1.5) |
|
Scrotal furunculosis/atheroma |
7 (1.5) |
|
Inguinal hernia |
6 (1.3) |
|
Symptomatic hydrocele |
5 (1.1) |
|
Varicocele |
4 (0.9) |
Twenty-nine percent (128) of patients presented with attendant symptoms [Table 2]. Ten percent (48) of patients underwent emergency surgery because testicular torsion was suspected. This was confirmed in 87.5% (42/48) of these patients, but 12.5% (6/48) of patients turned out to have acute epididymitis. Thirty-three percent (14/42) of patients with testicular torsion had a positive Prehn’s sign at presentation, 19.0% (8/42) had no cremaster reflex, and 54.7% (23/42) had a retained testis unilaterally.
Seventy-three percent (14/19) of patients presenting with testicular cancer exhibited an enlargement of the testis with only mild symptoms (pain). Still, they were triaged as acute scrotum, mainly due to the huge enlargement of the scrotum. In all patients for whom testicular cancer was either suspected or could not be ruled out, an ultrasound examination was performed. The ultrasound examination in 4.3% (19) of patients strongly indicated testicular cancer, and each of these cases was confirmed intraoperatively. Histological investigation showed testicular seminoma in 21.0% (4/19) and non-seminomatous germ cell tumor in 78.9% (15/19) of these patients. Eighty-nine percent (17/19) of patients with testicular cancer exhibited pathological tumor markers (β-subunit of human chorionic gonadotropin and/or α-fetoprotein).
Table 2: Attendant symptoms in patients (n = 128) presenting at the emergency department with acute scrotum.
|
Dysuria |
47 (36.7) |
|
Fever |
42 (32.8) |
|
Nausea/emesis |
15 (11.7) |
|
Lower abdominal pain |
11 (8.5) |
|
Pain radiation to the groin |
10 (7.8) |
|
Hematospermia |
8 (6.2) |
|
Macrohematuria |
6 (4.6) |
*Some patients had multiple symptoms.
Of the 58.4% (257) of patients with genital/paragenital infection, 90.2% (232/257) exhibited painful palpation, 49.0% (126/257) enlargement and 35.0% (90/257) swelling of the epididymis, and 15.1% (39/257) were hospitalized for further treatment.
All 42 patients with testicular torsion suffered from acute and increasing pain, which started ≤ 6 hours before their arrival at the ED. Of patients with genital/paragenital infection, 93.3% (240/257) presented with pain which started ≤ 6 hours before presentation at the ED, whereas only 6.6% (17/257) had pain for > 6 hours.
In 66.5% (293/440) of patients, both laboratory examinations and urinalysis were performed. Elevated CRP (> 3 mg/L) was found in 39.5% (116/293) of these patients, including 32.2% (83/257) of patients with genital/paragenital infection, 21.4% (9/42) of patients with testicular torsion, and 47.3% (9/19) of patients with testicular cancer. A pathological WBC count (> 10.4 g/L) was observed in 28.0% (72/257) of patients with genital/paragenital infection, 26.1% (11/42) of patients with testicular torsion, and 31.5% (6/19) of patients with testicular cancer. Six percent (19/293) of patients examined presented with both pathological laboratory findings (CRP > 3 mg/L; WBC > 10.4 g/L) and pathological urinalysis (> 4 WBC per field); 89.4% (17/19) of these patients had genital/paragenital infections, whereas 10.5% (2/19) had testicular torsion [Figure 1].
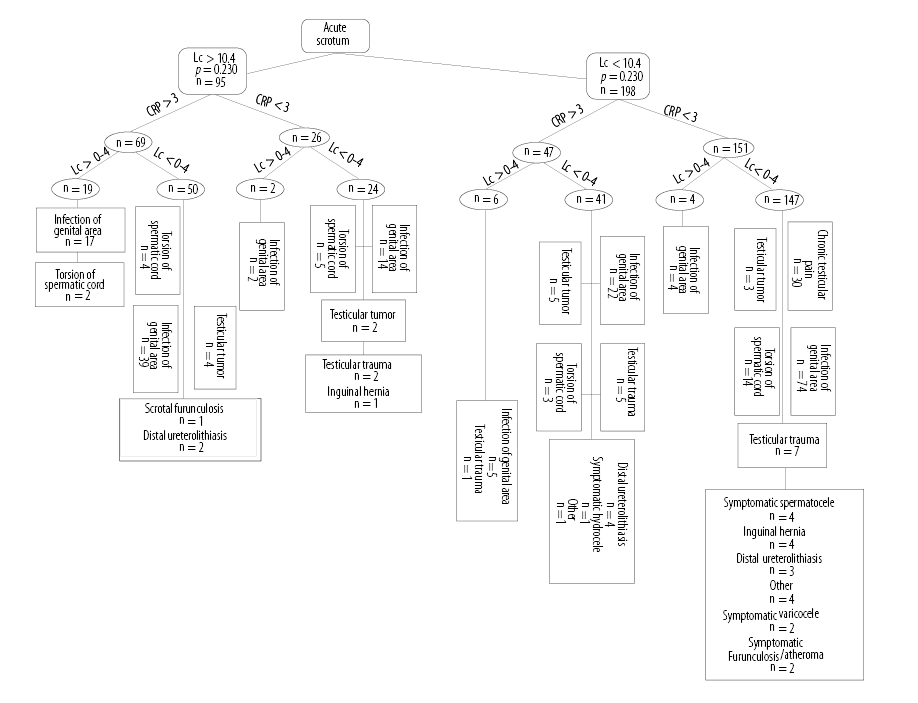
Figure 1: Diagnostic tree: The differential diagnosis according to the different laboratory parameters (blood leucocyte count (Lc), C-reactive protein (CRP), leucocyturia).
Ultrasound was performed in 86.5% (220/257) of patients with genital/paragenital infection. Fifty percent (110/220) of these patients had a pathological epididymis (enlargement) upon ultrasound examination and 46.3% (51/110) of these presented with additional hydrocele. Color Doppler ultrasound was performed in 47.6% (20/42) of patients with testicular torsion. In 65.0% (13/20) of these patients, pathological testicular perfusion was observed. The trigger for ultrasound was to rule out testicular tumor and/or epidydimitis if the symptoms were clinically unclear; Doppler ultrasound was performed if the symptoms and history pointed to inflammation (e.g., epidydimitis).
Table 3: Multivariate analysis showing the odds ratio (OR) with 95% confidence interval (CI) for each of the variables relevant to predicting the correct diagnosis.
|
CRP > 3 (blood) |
3.547 (1.254–4.258) |
0.025 |
|
0.780 |
|
WBC > 10.4 (blood) |
5.548 (2.761–12.587) |
0.023 |
|
0.120 |
|
WBC > 4 (urine) |
7.257 (4.269–14.589) |
0.002 |
|
0.170 |
|
Fever |
5.028 (1.929–13.103) |
0.001 |
|
0.970 |
|
Dysuria |
5.652 (2.18–14.655) |
0.004 |
|
0.740 |
|
Pain characteristics |
|
|
|
|
|
Burning |
0.537 (0.327–0.885) |
0.015 |
|
0.980 |
|
Stabbing |
2.731 (1.772–4.209) |
0.001 |
95.976 (12.797–719.807) |
0.001 |
|
Pain spreading |
|
0.050 |
|
0.630 |
|
Positive Prehn’s sign |
|
0.110 |
5.941 (1.432–24.658) |
0.014 |
CRP: C-reactive protein; WBC: white blood cells; ED: emergency department.
In multivariate regression analysis, a pathological WBC count in blood and/or urine, elevated CRP, dysuria, fever, and burning pain were independent and statistically significant predictors of genital/paragenital infection [Table 3]. A positive Prehn’s sign was an independent predictor of testicular torsion, whereas color Doppler ultrasound was not [Table 3].
Discussion
Acute scrotal pain is a bag of surprises, as the hidden cause can vary from severe medical conditions, such as testicular torsion or a strangulated inguinal hernia, to benign causes such as symptomatic varicocele or hydrocele. In this retrospective study, we identified genital/paragenital infections (epididymitis, epididymo-orchitis, prostato-vesiculo-epididymitis) as the leading cause of acute scrotal pain (58.4%). Testicular torsion was the third leading cause affecting 9.5% of patients with acute scrotal pain. To avoid serious consequences (i.e., loss of testis) an accurate and fast diagnosis is mandatory within six hours of symptoms onset to decide whether surgery is needed. We demonstrated that no single factor can reliably predict the cause of an acute scrotum, but rather a combination of factors, including patient history (characteristics of pain, dysuria), clinical findings (fever, Prehn’s sign), and laboratory tests (CRP and WBC in blood, WBC in urine). Noteworthy, an excellent collaboration between the emergency team and urologists is essential. However, as shown in our study, the experience of the urologists involved is another important factor. Because in our department an experienced urologist must always be involved, this could be the reason why none of the testicular torsions were missed, and 87.5% (42/ 48) scrotal explorations turned out to be testicular torsions.
In a retrospective seven-year review, Lyronis et al,1 showed that the most common overall cause of acute scrotum in 140 boys presenting with scrotal pain was epididymo-orchitis (35%), followed by torsion of scrotal appendages. The most common cause in the subgroup of preschool-age boys was spermatic cord torsion (p < 0.020). In contrast to these data on pediatric patients, D’Andrea et al,2 identified in a two-year retrospective review an infection in 56% (72/128) of adult patients with testicular pain. Eight percent of patients were diagnosed with testicular torsion, 6% with testicular trauma, 14% with a varicocele, and 16% with a hydrocele. These data are in line with our findings that approximately one in 10 adult patients with acute scrotum turns out to have testicular torsion.
Epididymitis is classically described as a slowly increasing painful process, in contrast to the rather acute onset of testicular torsion. However, making a clinical distinction between these two different causes of acute scrotum is often anything but obvious. The symptoms of patients with epididymitis range from local swelling and tenderness of the epididymis to massive erythema of the scrotum. Other attendant symptoms may be urethral discharge, dysuria, fever, and other lower urinary tract symptoms such as urethral burning.3 Ultrasonic examination of patients with epididymitis can show dilatation of the epididymis and an accompanying hydrocele, as well as increased blood flow in the inflamed area.2 In our study, only 15.5% (40/257) of patients with infection had pathological findings in the urine, which is in line with several published studies that reported pathological urine in 20% to 30% of patients with infection in the genital area.4,5 Mironov et al,6 conducted a laboratory study in 243 patients to establish the etiology of acute epididymitis. They found that acute epididymitis in sexually active young patients was caused primarily by sexually transmitted infections. However, urinary tract infection does not exclude testicular torsion, as two of the 42 patients with torsion in our cohort had > 4 WBC per field in urine.
In the present study, 9.5% (42/440) of patients with acute scrotum suffered from testicular torsion. In such cases, emergency surgical exploration must be performed as soon as possible, as irreversible ischemic injury to the testicular parenchyma may start to develop four to six hours after occlusion of the cord. As intratesticular blood flow was permanently absent in only 30% of these patients, there is no reliable, non-invasive diagnostic tool (not even color Doppler ultrasound), which is why surgical exploration is essential. Torsion can occur within or outside the tunica vaginalis in different age groups. Cases in neonates and children under 10 are rare but become more common in the second decade. This condition usually occurs in adolescents with fully descended testes and is often associated with an underlying structural malformation. In their meta-analysis, Hadway and Reynard7 found in patients suffering from testicular torsion a viable testis in 98% of patients with symptoms that lasted up to six hours at the time of surgery. Testicular viability dropped to 95% when surgery was performed within 12 hours and to < 89% when the patients underwent surgery within seven to 12 hours. The testis was saved in only 25% of patients when surgery occurred later than 12 hours.7 In another study covering a 16-month period and a total of 104 patients with torsion; no single clinical finding was found to exclude testicular torsion. The clinical features (pain < 24 hour, nausea/vomiting, abnormal cremasteric reflex, high position of the testis, positive Prehn’s sign) had 100% sensitivity for the diagnosis of testicular torsion. The clinical scoring system proved to be reliable, and reduced the negative exploration rate by > 55%. Ultrasound predictors alone were not able to identify all males with testicular torsion.8 Kalfa et al,9 reported a sensitivity of 89.9%, a specificity of 98.8%, and a false-positive rate of 1% for color flow Doppler ultrasound in the diagnosis of testicular torsion. In the early phase of torsion (1–3 hours), the echogenicity of the testis appears normal. With progression, common findings include enlargement of the affected testis and increased or heterogeneous echogenicity. A definitive diagnosis of complete testicular torsion is made when blood flow is visualized on the normal side but is absent on the affected side. In patients with incomplete torsion, some arterial flow persists in the affected testis.2
Interestingly, in the present study, the time between onset of pain and presentation at the ED was not a factor that could distinguish between genital/paragenital infection and testicular torsion. While all patients with torsion presented in the ED within six hours of pain onset, there was still a high percentage of these patients with genital/paragenital infection (93.3%).
Conclusion
Acute scrotum is a bag of surprises that demands immediate diagnostic resolution. While 58.4% of patients presenting at the ED with acute scrotum turned out to have inflammation in the genital/paragenital area, 9.5% had testicular torsion demanding immediate surgical intervention. Because time matters in such patients due to testicular ischemia, a fast and reliable diagnostic tool is highly desirable. Unfortunately, there is none. The decision tree presented in this study, however, is a viable alternative approach,
which demands close cooperation between the ED doctor and the urologist; 87.5% of surgical explorations for suspected testicular torsion turned out to be real ischemic torsions and not a single torsion was missed.
Disclosure
The authors declared no conflict of interest. No funding was received for this study.
Acknowledgements
We thank Dr. Konstantinos Karakostas for statistical support.
references
- 1. Lyronis ID, Ploumis N, Vlahakis I, Charissis G. Acute scrotum -etiology, clinical presentation and seasonal variation. Indian J Pediatr 2009 Apr;76(4):407-410.
- 2. D’Andrea A, Coppolino F, Cesarano E, Russo A, Cappabianca S, Genovese EA, et al. US in the assessment of acute scrotum. Crit Ultrasound J 2013 Jul;5(Suppl 1):S8.
- 3. Youssef F.R, Shipstone D. The acute scrotum. Surgery - Oxford international Edition 2011 Jun;29(6):278-281.
- 4. Kim SD, Kim SW, Yoon BI, Ha US, Kim SW, Cho YH, et al. The relationship between clinical symptoms and urine culture in adult patients with acute epididymitis. World J Mens Health 2013 Apr;31(1):53-57.
- 5. Redshaw JD, Tran TL, Wallis MC, deVries CR. Epididymitis: a 21-year retrospective review of presentations to an outpatient urology clinic. J Urol 2014 Oct;192(4):1203-1207.
- 6. Mironov VN. [Methods of laboratory studies in the etiology of acute epididymitis]. Urologiia 2003 Jul-Aug;(4):36-40.
- 7. Hadway PR, Reynard JM. The six-hour rule for testis fixation in testicular torsion: is it history? Journal of Clinical Urology 2013;6(2):84-88.
- 8. Boettcher M, Krebs T, Bergholz R, Wenke K, Aronson D, Reinshagen K. Clinical and sonographic features predict testicular torsion in children: a prospective study. BJU Int 2013 Dec;112(8):1201-1206.
- 9. Kalfa N, Veyrac C, Baud C, Couture A, Averous M, Galifer RB. Ultrasonography of the spermatic cord in children with testicular torsion: impact on the surgical strategy. The Journal of urology 2004;172(4 Pt 2):1692-1695.