Diabetic striatopathy is an uncommon presentation of diabetes mellitus. It usually presents with hemichorea-hemiballismus . Chorea means ῾dance-like’ in Greek and refers to rapid, chaotic movements that seem to flow from one body part to another. Many patients have generalized chorea, but most have asymmetric manifestations, and up to 20% have hemichorea.1 Ballismus is a type of involuntary movement affecting proximal limb musculature, manifesting as jerky, and flinging movements of the extremities.
The most common etiology of chorea in the pediatric age group is Sydenham’s chorea.2 In patients with diabetes mellitus, hyperosmolar nonketotic hyperglycemia is responsible for hemichorea-hemiballismus, which is commonly seen in type 2 diabetic adults, female patients in the hyperglycemic hyperosmolar state,3 and very few cases have been reported in the pediatrics age group. Hemichorea-hemiballismus movements associated with nonketotic hyperosmolar hyperglycemia in diabetic patients are labeled as diabetic striatopathy.4 It results from vascular and metabolic injury to basal ganglia due to chronic hyperglycemia resulting from poorly controlled type 2 and rarely type 1 diabetes mellitus.
We hereby present two cases of diabetic striatopathy who presented with abnormal body movements.
Case reports
Case one
An 11-year-old female presented with abnormal movements of the left hand and left foot for one and a half months. The movements were present continuously throughout the day and disappeared at night during sleep. The patient had type 1 diabetes for three years and was on the neutral protamine hagedorn (NPH) insulin regimen with poor compliance. There was no family history of diabetes. Anthropometry revealed short stature 119/125 cm (< -3 SD height for age on Centers for Disease Control and Prevention (CDC) growth chart), underweight 19/20.3 kg (< -3 SD weight for age on CDC growth chart). There were continuous, irregular, high-frequency movements interrupted by jerks consistent with hemichorea-hemiballismus. Laboratory results revealed glycated hemoglobin (HbA1c) level of 13.8%, random blood sugar (RBS) of 300 mg/dL, urinary ketones negative, and glycosuria present. Blood osmolality was 300 mOsm/kg. Antibodies against glutamic acid decarboxylase (anti-GAD 65) were positive, which confirmed the diagnosis of type 1 diabetes mellitus. Anti-insulin antibodies and anti-tissue transglutaminase were both negative. Brain magnetic resonance imaging (MRI) done on day four of admission revealed bilateral basal ganglia hemorrhage and ischemic changes, as shown in Figures 1 and 2. Correction of hyperglycemia was done with titration of insulin dosage. For abnormal body movements, trihexyphenidyl was started at 0.1 mg/kg, and the dosage increased to 0.2 mg/kg for optimal response. The patient was discharged under satisfactory conditions after 10 days of hospitalization. This patient did not return for follow-up.

Figure 1: T1-weighted MRI showing hyperintensity in bilateral basal ganglia more on the right side.
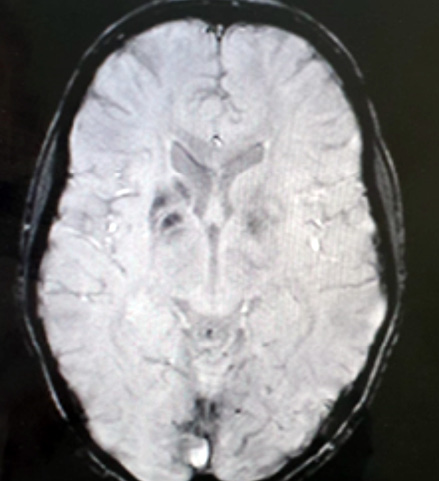
Figure 2: Gradient echo sequences showing blooming in basal ganglia right more than left due to hemorrhage.
Case two
A 10-year-old female presented with abnormal movements of the left upper and lower limbs for seven days. The patient had type 1 diabetes for two years was on NPH insulin regimen with poor compliance. There was no family history of diabetes mellitus. Anthropometry revealed short stature 110/123 cm (< -3 SD height for age on CDC growth chart) and that she was underweight 18/20.7 kg (< -3 SD weight for age on CDC growth chart). She had periodic choreiform and ballistic movements of the left upper and lower extremities that ceased during sleep. Initial laboratory values revealed RBS of 568 mg/dL and HBA1c of 10%. Urinary ketones were negative. Blood osmolality was 298 mOsm/kg. Brain MRI showed asymmetrical T1 hyperintensity in the bilateral basal ganglia (more on the right side) with no diffusion restriction post-contrast enhancement [Figure 3]. Anti-GAD 65 were positive, which confirmed the diagnosis of type 1 diabetes. Anti-insulin antibodies and anti-tissue transglutaminase were negative. Correction of hyperglycemia was done with titration of insulin dosage. For the abnormal body movements, she was started on trihexyphenidyl 0.1 mg/kg and clonazepam 0.015 mg/kg/day, and the dosage increased for optimal response (trihexyphenidyl 0.3 mg/kg/day, clonazepam 0.03 mg/kg/day, and haloperidol 0.015 mg/kg/day). She was discharged on a basal-bolus regimen. On follow-up, one month after discharge, the patient showed improved choreiform movements and glycemic control. The patient remained on regular follow-up (every three months) and showed improvements in dyskinetic movement. Treatment with haloperidol and trihexyphenidyl tapered and stopped.
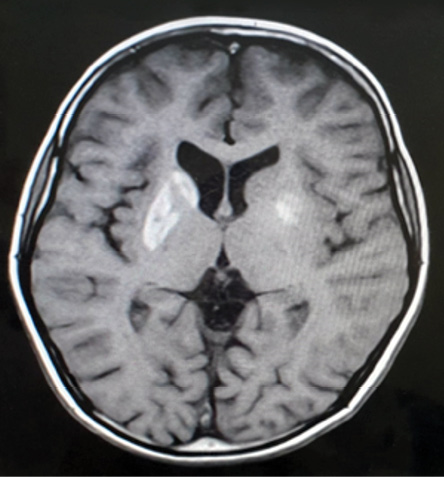
Figure 3: Bilateral hyperintensity in basal ganglia more on the right side.
Electroencephalogram was not done in both cases as after MRI images, the diagnosis of diabetic striatopathy was made, and patients improved with treatment.
Weight and height were mentioned for the patient as a part of the general physical examination and to see nutritional adequacy on follow-up.
Discussion
Diabetic striatopathy presents with hemichorea-hemiballismus in patients with poorly controlled hyperglycemia. Chronic hyperglycemia causes vascular and metabolic injury to basal ganglia causing contralateral hyperkinetic movements. The neuroimaging shows lesions, such as hyper attenuation of basal ganglia on computed tomography scan, the hyperintensity of basal ganglia on MRI (T1-weighted imaging).
The clinical findings in our cases are consistent with striatopathy due to nonketotic hyperglycemia. The exact etiopathogenesis of diabetic striatopathy and non-ketosis is not fully understood. Two major hypotheses have been purposed to explain the likely etiology: ischemic versus metabolic insult.5
The difference in the incidence of striatopathy in type 2 diabetic patients compared to type 1 is due to pathophysiology of dyskinesia. In hyperglycemic hyperosmolar state (HHS), the Krebs cycle activity is suppressed in the brain due to hyperglycemia and shifts to anaerobic cycle. This shift in HHS causes the brain to metabolize gamma-Aminobutyric acid (GABA) to succinic acid via the semialdehyde pathway, hence depleting GABA levels in the brain. This leads to the release of inhibitory control by GABA in the thalamus resulting in hyperkinetic movement, whereas in diabetic ketoacidosis (DKA) acetoacetate produces in the liver and GABA levels are deranged, hence the rarity in DKA patients.6,7 The nonketotic hyperglycemia suppresses insulin secretion, which leads to impaired glucose transport in brain cells. Impaired glucose transport leads to diminished functioning of GABAnergic inhibitory transmission in the striatum, which manifests clinically as abnormal body movements. The metabolic hypothesis does not explain why after good glycemic control movements cease to disappear in a few patients.8,9
The ischemic hypothesis suggests that regional ischemia is due to diabetic vasculopathy. Under the effect of hyperviscosity, which is induced by dehydration due to nonketotic hyperglycemia, a thrombotic obstruction of vessels or transient ischemia is liable to occur. Although obstruction of the microvascular lumens in the affected striatum has seldom been observed histologically.8,9
In most cases of diabetic striatopathy, hypergly-cemia is usually present without ketoacidosis, as seen in our patients too, which explains the fact that these patients tolerate persistent hyperglycemia leading to changes in basal ganglia. The exact reason behind the basal ganglia lesion is still unclear. In typical hemiballismus-hemichorea patients, MRI findings of hyperintensity in basal ganglia are due to the accumulation of manganese in gemistocytes (astrocytes with large protein content found in acute brain injury). Histopathological findings in adult diabetic patients with hemiballismus-hemichorea revealed areas of gliosis, accumulation of gemistoctyes, and loss of neurons. The majority of data in diabetic hemiballismus-hemichorea is on adult patients. As few as three pediatric patients have been reported. Hence, more studies are required in pediatric patients who present with non-ketotic hyperglycemia.10,11
Most patients with hemichorea-hemiballismus improve with improved glycemic control and neuroleptic drugs. However, the exact duration of anti-chorea treatment is not known. The most common drugs used for choreiform movements are neuroleptics drugs (haloperidol/risperidone), selective serotonin reuptake inhibitor (escitalopram), dopamine depleting agent (risperidone tetrabenazine), and GABAnergic drugs (clonazepam/gabapentin). The complete clinical improvement with or without radiological improvement may take from two to 28 days.3 The rationale for using trihexyphenidyl in our cases is due to its antidyskinetic effect.12 The mean time of improvement in case one was 30 days, and three months in case two.
Conclusion
Close monitoring is required in patients who have hyperglycemia without ketosis. These patients have more tendencies to develop hemichorea-hemiballismus, and early institution of therapy and glucovigilance in these patients can prevent long-term morbidity.
Disclosure
The authors declared no conflicts of interest. The consent from parents was taken before collecting the data for both cases.
Acknowledgements
Department of Radiology, Guru Gobind Singh Medical College, Faridkot, Punjab, India.
references
- 1. Rebbeca K, Jonathan W. Chorea, athetosis, Tremor. In: Kliegman R.F, Stanton B.F, Schor N.F, Geme J.W.St, Behrman R.E.(Eds). Nelson Textbook of Pediatrics. 19th edition.Philadelphia,USA: Elsevier; 2011. p. 2888 -2891.
- 2. Davico C, Canavese C, Tocchet A, Brusa C, Vitiello B. Acute Hemichorea Can Be the Only Clinical Manifestation of Post-Varicella Vasculopathy: Two Pediatric Clinical Cases. Front Neurol 2018 Mar;9:164.
- 3. Das L, Pal R, Dutta P, Bhansali A. “Diabetic striatopathy” and ketoacidosis: Report of two cases and review of literature. Diabetes Res Clin Pract 2017 Jun;128(128):1-5.
- 4. Lucassen EB, Delfyett WT, Stahl MC. Persistent hemichorea and caudate atrophy in untreated diabetic striatopathy. Case Rep Neurol 2017 Dec;9(3):299-303.
- 5. Ohara S. Diabetic Hemichorea-hemiballism. Austin J Clin Neurol 2015;2(4):1037.
- 6. Lin JJ, Chang MK. Hemiballism-hemichorea and non-ketotic hyperglycaemia. J Neurol Neurosurg Psychiatry 1994 Jun;57(6):748-750.
- 7. Mihai CM, Catrinoiu D, Stoicescu RM. Atypical onset of diabetes in a teenage girl: a case report. Cases J 2008 Dec;1(1):425.
- 8. Tiwari V, Ambadipudi S, Patel AB. Glutamatergic and GABAergic TCA cycle and neurotransmitter cycling fluxes in different regions of mouse brain. J Cereb Blood Flow Metab 2013 Oct;33(10):1523-1531.
- 9. Pisani A, Diomedi M, Rum A, Cianciulli P, Floris R, Orlacchio A, et al. Acanthocytosis as a predisposing factor for non-ketotic hyperglycaemia induced chorea-ballism. J Neurol Neurosurg Psychiatry 2005 Dec;76(12):1717-1719.
- 10. Ohara S, Nakagawa S, Tabata K, Hashimoto T. Hemiballism with hyperglycemia and striatal T1-MRI hyperintensity: an autopsy report. Mov Disord 2001 May;16(3):521-525.
- 11. Faundez T, Klee P, Hanquinet S, Schwitzgebel V, Burkhard PR, Korff CM. Diabetic striatopathy in childhood: a case report. Pediatrics 2016 Apr;137(4):e20143761.
- 12. Wate SP, Borkar AA. Simultaneous spectrophotometric estimation of haloperidol and trihexyphenidyl in tablets. Indian J Pharm Sci 2010 Mar;72(2):265-267.